- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
Infertility is defined as “the failure to conceive after one year or more of frequent, unprotected sexual intercourse.” It is estimated that 1 in 7 couples are affected by infertility in the UK.1
80% of couples trying to conceive will do so within 1 year. Half of the couples who did not conceive in the first year will conceive in the second, resulting in an overall pregnancy rate in 2 years of 90%.1
Infertility can be classed as:
- Primary – couples who have never conceived
- Secondary – couples who have previously conceived
Hypothalamic-pituitary-gonadal (HPG) axis
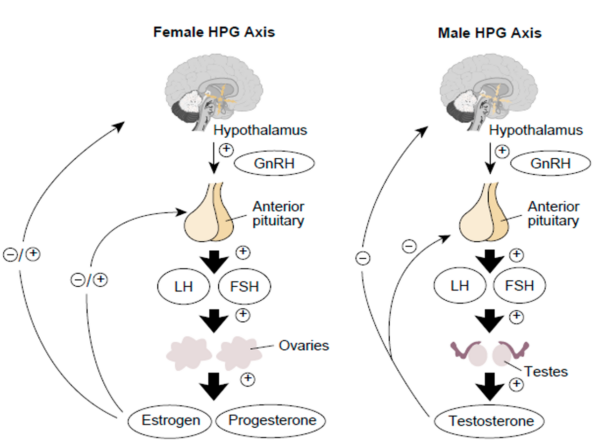
To fully understand the topic of infertility and actions of infertility drugs, it is useful to have an overview of the hypothalamic-pituitary-gonadal (HPG) axis (Figure 1).
The hypothalamus releases gonadotrophin-releasing hormone (GnRH). GnRH acts on the anterior pituitary to stimulate the secretion of follicle-stimulating hormone (FSH) and luteinising hormone (LH).
In females, LH and FSH then stimulate the ovaries to produce progesterone and oestrogen. When there are moderate levels of oestrogen, or oestrogen is secreted in the presence of progesterone, negative feedback on the HPG axis inhibits GnRH release.
However, when there are high levels of oestrogen present, positive feedback on the HPG axis stimulates the hypothalamus to secrete GnRH.
In males, LH acts on Leydig cells within the testes to produce testosterone. Testosterone and FSH then interact with Sertoli cells to stimulate sperm development. Testosterone also provides negative feedback to the hypothalamus to suppress GnRH secretion.
Causes of infertility
The main causes of infertility in the UK are:
- Male factor infertility (30%)
- Ovulatory causes for infertility (25%)
- Tubal causes for infertility (20%)
- Uterine/peritoneal causes for infertility (10%)
In 40% of affected couples, both male and female infertility factors are responsible for infertility.1
Female factor infertility
Causes of female factor infertility are commonly separated into:
- Disorders of ovulation
- Tubal causes
- Uterine/peritoneal causes
Disorders of ovulation
To understand pathological processes that can affect ovulation, it is important to know the basics of the menstrual cycle (read our guide here). There are 3 main types of ovulatory disorders.
Group I – hypothalamic-pituitary failure
- Hypogonadotropic hypogonadism: There is a failure to produce the required amount of LH and FSH, resulting in anovulation.
Group II – hypothalamic-pituitary-ovulation dysfunction
- This occurs as the result of polycystic ovary syndrome (PCOS) – the most common cause of female infertility.
Group III –ovarian failure
- Hypergonadotropic hypogonadism: There is normal hypothalamic and pituitary function but there are insufficient numbers of follicles within the ovary. Therefore, there is less oestrogen produced and follicles do not develop fully. This results in anovulatory cycles.2
Other ovulatory causes include:
- Sheehan’s syndrome – Hypopituitarism caused by ischaemic necrosis of the pituitary. This occurs as the result of severe hypotension or haemorrhagic shock secondary to massive post-partum haemorrhage.
- Hyperprolactinaemia – This inhibits both FSH and LH secretion and can lead to menstrual dysfunction and galactorrhoea.
- Pituitary tumours – The tumour displaces or destroys normal pituitary tissue and can affect the production of FSH and LH.2
Tubal causes
The delicate structure of the fallopian tubes makes them more susceptible to damage. The most common cause of tubal damage is due to pelvic inflammatory disease, which is usually secondary to chlamydia or gonorrhoea infection.
Other tubal causes include:
- Previous sterilisation
- Endometriosis
- Previous pelvic surgery
Uterine/peritoneal causes
The most prevalent uterine or peritoneal cause is endometriosis, which causes inflammation and adhesions in the pelvis that can distort pelvic anatomy.
Other uterine/peritoneal causes include:
- Cervical mucus dysfunction or defect
- Previous pelvic or cervical surgery
- Uterine fibroids
- Asherman’s syndrome
- Previous abdominal infections which have resulted in adhesions (e.g. appendicitis)
- Congenital abnormalities
Male factor infertility
Traditionally, male factor infertility is diagnosed by observing sperm abnormalities during semen analysis. Semen analysis assesses sperm count, motility, morphology, vitality, concentration and volume.
Potential abnormalities on semen analysis include:
- Oligospermia – < 15 million sperm per ml
- Teratospermia – < 4% normal morphology
- Asthenospermia – < 32% sperm motility
- Azoospermia – no sperm found within the ejaculate1,3
Male factor infertility can be classified into:
- Obstructive infertility – a problem with the sperm delivery
- Non-obstructive infertility – a problem with the sperm production
- Coital infertility – infertility secondary to sexual dysfunction4
Obstructive infertility
- Previous vasectomy – the most common cause of obstructive azoospermia
- Cystic fibrosis – congenital absence of the vas deferens
- Ejaculatory duct obstruction – can be caused by previous prostatitis, leading to fibrosis of the ejaculatory duct or by congenital prostatic cysts
- Epididymal obstruction – may occur secondary to a chlamydia or gonorrhoea infection which can cause inflammation and fibrosis of the epididymis
Non-obstructive infertility
- Hormonal causes- hypogonadotropic hypogonadism, hyperprolactinaemia – causes infertility secondary to impotence
- Varicocele – is associated with impaired testicular function and infertility
- Genetic causes:
- Klinefelter’s syndrome (47, XXY) – This is the most common genetic disorder associated with infertility. It is associated with hypogonadism and defects of spermatogenesis.
- Androgen insensitivity syndrome – Child with XY karyotype appearing female due to resistance of the virilising actions of androgens. When there is limited resistance to androgens, there may be poor development of the testes.
- Kallmann syndrome – This is a form of hypogonadotropic hypogonadism. Males affected by this genetic disorder usually have undescended testes and a micropenis. The lack of sex hormone production often causes a lack of development of secondary sexual characteristics. 3
- Cryptorchidism (undescended testes)
- Previous testicular trauma or damage (e.g. testicular torsion)
- Testicular malignancy
Coital infertility
- Erectile dysfunction – when severe, there may be difficulties with semen deposition
- Premature ejaculation – when severe, ejaculation may occur before vaginal penetration
- Anejaculation:
- Primary – may occur due to psychosexual or neurological causes
- Secondary – may occur due to previous abdominal/pelvic surgery or certain drugs such as antidepressants or alpha-blockers
- Retrograde ejaculation
- Penile deformities (e.g. Peyronie’s disease, hypospadias)
Investigations
Investigations for infertility may be commenced in couples who have been trying to conceive for 1 year after frequent (every 2-3 days), unprotected sexual intercourse.
Couples can have an early referral for infertility investigations, after 6 months of unsuccessful attempts to conceive, if they meet the following criteria:
- The woman is aged 36 years or older or
- There is a known cause of infertility or
- There is a history of predisposing factors
*Immediate assessment and treatment of couples may be warranted when the woman is aged 40 years or older.1
Initial investigations
These investigations may be requested in primary care.
Male
- Semen analysis – this assesses the sperm count, motility, morphology, vitality, concentration and volume.
- Chlamydia screen3
Female
- Mid-luteal progesterone (day 21 or equivalent in the woman’s cycle) – to assess whether the woman is ovulating
- FSH and LH to assess ovarian function – poor ovarian function may be indicated by high levels of both FSH and LH
- Chlamydia screen2
Further investigations
These are often conducted in secondary care.
Male
- Hormone analysis – testosterone, FSH, LH and prolactin
- Genetic testing
- Ultrasound – to investigate any potential structural abnormality
- Testicular biopsy – to both define any histological diagnosis and potentially extract any sperm
- Viral screen – HIV, Hepatitis B and Hepatitis C screening should be offered to those undergoing IVF treatment.3
Female
- Investigations for tubal patency:
- Hysterosalpingogram (HSG)
- Laparoscopy and dye – usually offered when women are known to have a comorbid condition (e.g. endometriosis)
- Investigations of the ovarian reserve:
- One of the following should be measured on day 3 of the woman’s cycle to predict the ovarian response to gonadotrophin stimulation in IVF treatment:
- Total antral follicle count
- Anti-Mullerian hormone (AMH) (low count= premature ovarian failure)
- FSH
- Viral screen – HIV, Hepatitis B and Hepatitis C screening should be offered for people undergoing IVF treatment.2
- One of the following should be measured on day 3 of the woman’s cycle to predict the ovarian response to gonadotrophin stimulation in IVF treatment:
Management
Initial preconception lifestyle advice, which ideally would be given in primary care:
- Encourage regular intercourse – every 2-3 days
- 400 micrograms folic acid daily(take 5mg daily if high risk for neural tube defects e.g. diabetes, on anti-epileptics etc.)
- Smoking cessation
- Reduce alcohol intake if excessive – as known to impair fertility
- Optimise weight – obesity in women is associated with PCOS, miscarriage, lower success with assisted reproductive technologies and increased risk of obstetric complications. Being overweight can also impair fertility in males.
- Healthy diet and regular exercise
- Men to wear loose-fitting clothing
Female factor infertility
Medical management
Medication to stimulate ovulation. Consult your local BNF for appropriate dosages.
- Clomiphene – an anti-oestrogen drug:5
- Clomiphene induces ovulation by inhibiting oestrogen from binding in the anterior pituitary. This stops the negative feedback mechanism of oestrogen, thus the secretion of GnRH, FSH and LH increases. This results in greater stimulation of the ovaries and therefore a greater increase in oestrogen production and secretion. The oestrogen stimulates follicle growth and maturation.6
- Gonadotrophins – can be given when there is clomiphene-resistant anovulatory infertility
- Pulsatile GnRH:
- GnRH must be given in a pulsatile fashion to stimulate the release of gonadotrophins.
- GnRH given continuously actually has an inhibitory effect.
- Dopamine agonists – these can be beneficial when the ovulatory disorder is secondary to hyperprolactinaemia5
Surgical management
- Tubal surgery:
- Used for women with mild tubal disease.
- This includes tubal catheterisation or tubal cannulation to improve the chance of a spontaneous pregnancy.1,5
Male factor infertility
Medical management
- Gonadotrophins may be given in men with hypogonadotropic hypogonadism5
Surgical management
- Surgical correction of any blockage within the male genital tract should be offered to restore patency and improve fertility.1,5
Assisted conception methods for female and male factor infertility
Assisted conception refers to procedures which cause sperm to come into proximity with oocytes to promote conception. It includes:
- Intrauterine insemination (IUI) – this uses partner or donor sperm and is inseminated during natural or stimulated cycles
- In vitro fertilisation (IVF) – this method is beneficial for women who have more severe tubal disease when there is unexplained infertility or they have found other treatments to be unsuccessful
- Intracytoplasmic sperm injection (ICSI) – this procedure involves injecting a sperm directly into an oocyte. This is a suitable option for men with a low sperm count or with sexual dysfunction.
- Donor insemination – this may be used when the man suffers from persistent azoospermia and there are very few or no sperm identified on a testicular biopsy
- Oocyte donation – this is a suitable option when the woman has ovarian failure or there is an absence of the ovaries
- Embryo donation 1,5
Complications of assisted conception
These can include:
- Ovarian hyperstimulation syndrome (OHSS) – this is a potentially life-threatening complication of superovulation
- Multiple pregnancy
- Ectopic pregnancy
- Pelvic infection1
Psychological support for couples with infertility
The diagnosis of infertility is associated with significant psychological stress. Whilst there are many treatment options to help couples affected by infertility, these treatments are not 100% effective. Therefore, it is important to evaluate and consider a couple’s mental health throughout this process.
NICE recommends offering counselling for patients who are diagnosed with fertility problems. It is also recommended that counselling should be offered before, during and after any investigation and treatment, regardless of the outcome of the procedures.1
References
- NICE Guideline CG156. Fertility problems: assessment and treatment. Published in 2013. [LINK]
- Smith S, Pfeifer S, Collins J. Diagnosis and Management of Female Infertility. JAMA. 2003;290(13):1767.
- Karavolos S, Stewart J, Evbuomwan I, McEleny K, Aird I. Assessment of the infertile male. The Obstetrician & Gynaecologist. 2013;15(1):1-9.
- Abdel Raheem A, Ralph D, Minhas S. Male infertility. British Journal of Medical and Surgical Urology. 2012;5(5):254-268.
- Nandi A, Homburg R. Unexplained subfertility: diagnosis and management. The Obstetrician & Gynaecologist. 2016;18(2):107-115.
- Rang, H.P. & Dale, M. Maureen.Rang & Dale’s Pharmacology. 7th ed. Edinburgh: Elsevier Churchill Livingstone; 2012. [LINK]
- Lu Kong, Ting Zhang, Meng Tang and Dayong Wang. Female HCG axis. [CC BY] [LINK]
- Lu Kong, Ting Zhang, Meng Tang and Dayong Wang. Male HCG axis. [CC BY] [LINK]
Reviewer
Dr Brenda Narice
Obstetrics & Gynaecology Registrar
Editor
Hannah Thomas




