- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Pancreatic cancer is a common and deadly disease; one of those nightmare cancers that tend to progress insidiously and unrecognisably until finally detected at a late and incurable stage. Despite massive advances in surgical and medical management over the last few decades, it continues to have a terrible prognosis and many patients die within months of diagnosis. It is vital for doctors at all stages of training to have an awareness of the symptoms and appropriate initial investigations in order to have the best chance of identifying pancreatic cancer early, when surgeons might still be able to remove it. The majority of pancreatic cancers are ductal adenocarcinomas, so this article will focus mainly on these.
Anatomy and physiology of the pancreas
Anatomy
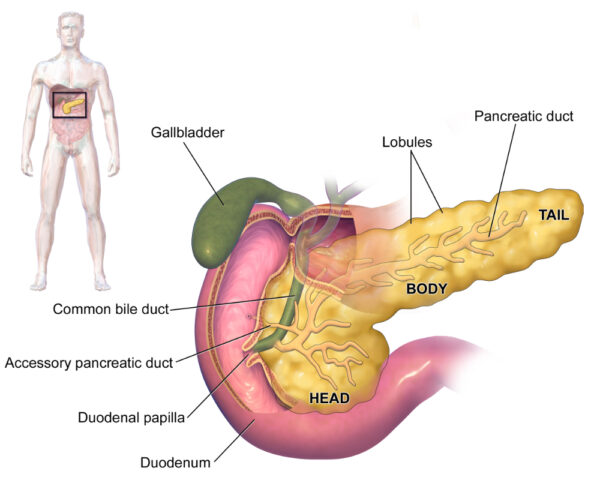
- the pancreas is a soft, spongy, lobulated organ which sits in the upper part of the retroperitoneal space of the abdomen
- it lies behind the stomach and transverse colon, and the lesser sac
- it sits in front of the spinal column, abdominal aorta, inferior vena cava, portal vein, left kidney and spleen
- due to its position behind several organs and beneath the ribs, it is not usually palpable on abdominal examination
- the pancreas has four parts:
- a head – this fits nicely into the C-shaped cavity created by the duodenum and usually has a small uncinate process which hooks upwards behind the superior mesenteric artery (SMA) and superior mesenteric vein (SMV)
- the constricted neck connects the head to the body – behind this, the portal vein forms from the union of the splenic and superior mesenteric veins
- the body runs upwards and to the left across the midline
- the tail travels with the splenic vessels between the layers of the splenorenal ligament, to reach the hilum of the spleen
- the main pancreatic duct (of Wirsung) begins in the tail of the pancreas and runs along the length of the gland before joining the common bile duct at the ampulla of Vater. It then drains into the second part of the duodenum at the major duodenal papilla. Some people have an accessory pancreatic duct (of Santorini) which drains the upper part of the pancreatic head and opens into the duodenum at the minor duodenal papilla.
- the pancreas receives its arterial blood supply from several vessels:
- the head is supplied by the superior pancreaticoduodenal artery (from the gastroduodenal artery) and the inferior pancreaticoduodenal artery (from the superior mesenteric artery)
- the neck, body and tail are supplied by branches of the splenic artery (from the coeliac trunk) the largest of which is called the arteria pancreatica magna or greater pancreatic artery
- venous blood from the pancreas travels along corresponding veins to drain into the portal vein
- its lymphatics follow the course of the arteries to the coeliac and superior mesenteric lymph nodes
- its nerve supply is via an autonomic pancreatic plexus and consists of sympathetic trunk branches originating from T6-T10 and parasympathetic nerve fibres from the vagus nerve
Physiology
- the pancreas functions as both an exocrine and an endocrine gland
- exocrine glands produce secretions which are excreted via ducts onto a body surface, such as the skin or gastrointestinal (GI) tract – other examples include the salivary glands, breasts, lacrimal glands, and sebaceous and sweat glands in the skin
- endocrine glands produce hormones which are released into the bloodstream and travel in the circulation to distant target organs and tissues – other examples include the thyroid and parathyroid glands, adrenals, pituitary gland and pineal gland
- other organs with dual exocrine and endocrine function include the liver, kidneys and the gonads
- the exocrine pancreas consists of acinar and ductal cells which produce pancreatic juice; this is secreted into the duodenum via the pancreatic duct and aids the processes of digestion and absorption of food
- ductal cells produce bicarbonate solution which helps neutralise acidic stomach contents
- acinar cells produce a range of pancreatic digestive enzymes which break down complex food molecules into simpler ones so they can be absorbed by the small intestine – these include amylase, lipase, trypsinogen, chymotrypsinogen and carboxypeptidase
- the release of pancreatic juice is stimulated by duodenal secretin and cholecystokinin secretion, as well as gastrin, and inhibited by somatostatin and glucagon
- the endocrine pancreas consists of about a million tiny clusters of endocrine cells called the islets of Langerhans, which are embedded throughout the pancreatic tissue and have a combined weight of only 1.5 grams; there are several different cell types which release specific hormones into the bloodstream via capillaries and play an essential role in regulating glucose homeostasis and gut function:
- alpha (α) cells produce glucagon, which raises blood sugar levels
- beta (β) cells produce insulin, which lowers blood sugar levels
- delta (Δ) cells produce somatostatin, an inhibitory compound which suppresses the release of other gut hormones and decreases GI tract function
- PP (F or γ) cells produce pancreatic polypeptide, which regulates pancreatic secretions and may play a role in producing feelings of satiety
- epsilon (ε) cells produce ghrelin, which triggers feelings of hunger
- G cells produce gastrin, which stimulates gastric acid secretion and gastric motility
- other hormones which may be produced in the pancreatic islets include vasoactive intestinal peptide (VIP) and serotonin (5-HT)
- pancreatic failure may be exocrine (resulting in malabsorption, steatorrhoea and malnutrition), endocrine (resulting in diabetes) or both, and can be a presenting feature of pancreatic cancer
Epidemiology
- pancreatic cancer accounts for 2.6% of cancers in the UK (about 8700 cases per year)
- overall, it is the tenth most common cancer and causes 5.3% of cancer deaths (about 8600 a year)
- in men, it is the thirteenth most common cancer and has a lifetime risk of 1.4%, or 1 in 73
- in women, it is the ninth most common cancer and has a lifetime risk of 1.4%, or 1 in 74
- it has a peak incidence in the over 75s and is rare under the age of 40
- overall incidence has remained static since the mid-1970s – up until recently it had been decreasing, probably due to more people giving up smoking, but it has subsequently started to rise again, probably due to the increase in obesity and other lifestyle factors
- pancreatic cancer is much more common in developed countries in Eastern Europe and North America
Aetiology and risk factors
- the aetiology of pancreatic cancer is poorly understood. 95% of tumours carry mutations in the K-RAS2 proto-oncogene, the activation of which leads to increased cell proliferation, loss of the normal response to apoptotic signals, dysplasia and ultimately cancer. In the pancreas, K-RAS mutations tend to cause the development of precancerous lesions called pancreatic intraepithelial neoplasia (PIN), which are present in up to a third of elderly people at post-mortem but will only lead to cancer in about 1% of cases. It is estimated that it takes up to 20 years from the time of the first mutation to develop full-blown disease. K-RAS is also implicated in a number of other cancers, including colorectal and lung cancers.
- it is hypothesised that about 40% of cases of pancreatic cancer are sporadic (i.e. due to random unlucky genetic mutations), with 50% being linked to environmental and lifestyle factors and 5-10% being caused by inherited genetic disorders
- increasing age is a non-modifiable risk factor – the older you get, the more likely to are to have accumulated enough genetic mutations to develop a cancer
- blood group A patients have a 40% higher risk than those with other blood types
- being tall is also mysteriously a risk factor
- environmental and lifestyle factors which are known to increase the risk of pancreatic cancer include:
- smoking or chewing tobacco is profoundly carcinogenic and is linked to about 30% of all cases of pancreatic cancer – smokers have a 2.2x risk of developing the disease, and unfortunately, the risk remains increased in people who switch to e-cigarettes (1.6x)
- obesity is linked to about 12% of cases. This is thought to be due to the hyperinsulinaemic state generated by the metabolic syndrome, which promotes increased pancreatic cell division to meet demands.
- excessive alcohol consumption (>6 drinks per day) is also implicated in a number of cases and can increase risk by up to 1.6x. If you have less than 3 drinks per day, the evidence suggests you’re safe.
- a high intake of red or processed meats has also been implicated in men
- ionising radiation (e.g. from repeated CT scans or radiotherapy for previous cancers) is a well recognised environmental risk factor linked to 2-4% of cases
- occupational exposure to acrylamide (used in dyes, fabrics and paper manufacturing) leads to 0.01% of cases
- pancreatic cancer has also been linked with a number of other medical conditions:
- diabetes mellitus (especially if on gliclazide) – 2x risk
- chronic pancreatitis – 3x risk
- hereditary pancreatitis – 50x risk
- gallstones – 1.25x risk
- previous cholecystectomy – 1.23x risk
- previous gastrectomy – 6x risk
- chronic infections – hepatitis B, H.pylori, chronic dental problems
- pretty much any other cancer – this may indicate shared causative risk factors or the effects of chemoradiotherapy leading to a secondary malignancy
- family history and inherited genetic disorders are also very important:
- any patient with an affected first-degree relative has a 1.7x increase in risk
- Peutz-Jegher’s syndrome – 100x risk
- familial atypical multiple mole melanoma syndrome (FAMMM) – about 30x risk
- hereditary non-polyposis colon cancer (HNPCC) – 9x risk
- BRCA2 – 3.5x risk
- BRCA1 – 2.3x risk
- MEN1 causes pancreatic endocrine tumours in 50% of cases
- von-Hippel Lindau syndrome causes pancreatic cystic lesions which become malignant in 17% of cases
- possible protective factors include having allergies, exercise, high folate diet, citrus fruits and nuts
Location
The anatomical location of a pancreatic tumour is important in dictating its likely presenting symptoms.
- 65% head/neck of the pancreas – these tend to present earlier with obstructive jaundice
- 15% body/tail of the pancreas – these tend to present later and have a worse prognosis
- 15% diffuse or multicentric involvement of the whole pancreas
Histopathological types of pancreatic cancer
95% EXOCRINE TUMOURS – these are derived from the ductal epithelium, acinar cells, connective tissues and lymphatics; they are by far the most common type and may be solid or cystic:
- solid exocrine tumours include:
- 90% pancreatic ductal adenocarcinoma: subtypes of this include mucinous non-cystic carcinoma, adenosquamous carcinoma, giant cell carcinoma and anaplastic carcinoma
- 5% acinar cell carcinoma
- <1% pancreatoblastoma: these are very rare embryonal tumours, which tend to present as a large abdominal mass in children aged 1-15 years
- <5% other solid tumours: these would include lymphomas, teratomas, and metastases from other sites e.g. GI tract, kidneys, lung, breast and melanoma
- cystic exocrine tumours are less common but have a better prognosis than solid tumours; they include:
- cystadenocarcinoma: these may be subclassified as mucinous or serous
- intraductal papillary-mucinous neoplasm (IPMN): these are derived from the pancreatic ducts and may cause either localised cancer or diffuse malignant change throughout the pancreas
- pseudopapillary carcinoma
5% ENDOCRINE TUMOURS – these are derived from the cells of the islets of Langerhans and the neuroendocrine system; they are rare but affect up to 75% of patients with MEN1 syndrome
- one-third are non-functional – up to 90% of these are malignant, and as they do not cause hormonal symptoms they tend to present late with a large mass and/or metastatic disease
- some of these may produce pancreatic polypeptide, which helps with making the diagnosis but does not actually cause symptoms
- two-thirds are functional – overall about 50% of these are malignant, and they can cause a variety of symptoms and syndromes depending on which hormone they produce:
- insulinomas cause Whipple’s triad of symptomatic fasting hypoglycaemia which is relieved by glucose administration
- glucagonomas cause diabetes, weight loss and paraneoplastic phenomena such as thrombophlebitis migrans and nail changes
- gastrinomas cause Zollinger-Ellison syndrome, which is characterised by complex refractory peptic ulcer disease relieved by PPI administration, along with diarrhoea/steatorrhoea
- VIPomas cause Verner-Morrison syndrome, which consists of severe watery diarrhoea leading to weight loss, hypovolaemia and electrolyte imbalances, as well as nausea, flushing and cramps
- somatostatinomas are very rare and lead to diarrhoea/steatorrhoea, malabsorption, weight loss and electrolyte disturbances
Symptoms
Painless obstructive jaundice is due to cancer until proven otherwise
- 70% of patients present with progressive obstructive jaundice with dark urine and pale stools, due to a tumour in the head or neck of the pancreas extrinsically compressing the common bile duct
- the jaundice is usually painless but can be painful – the pain is similar to that of acute pancreatitis, in that it is usually epigastric, radiates through to the back and is relieved by sitting forward
- the onset of jaundice is often preceded by several months of non-specific anorexia, malaise, weight loss and vague abdominal or back pain
- infection of stagnant biliary fluid can lead to ascending cholangitis and sepsis
- less commonly, pancreatic cancer can present as unexplained acute pancreatitis or new-onset diabetes, malabsorption or steatorrhoea in an elderly person
- there may be features of paraneoplastic syndromes such as:
- hypercalcaemia
- acanthosis nigricans – skin fold hyperpigmentation
- thrombophlebitis migrans – recurrent, migratory superficial venous thrombophlebitis
- non-infective “marantic” endocarditis
- neuromuscular syndromes – dermatomyositis, polymyositis, Lambert-Eaton myasthenic syndrome
- symptoms of advanced disease include:
- nausea, anorexia, fatigue, malaise
- vomiting due to gastric outlet obstruction
- portal hypertension due to splenic vein thrombosis – this may present as an oesophageal variceal bleed with haematemesis or malaena
- symptoms of metastatic disease include RUQ pain (liver), shortness of breath and haemoptysis (lungs), neurological deficits or seizures (brain), abdominal distension and ascites (peritoneal spread) and subcutaneous nodules (skin)

Signs
- general examination may reveal jaundice, pallor, skin excoriations or weight loss/cachexia
- abdominal examination may reveal:
- epigastric tenderness
- an upper abdominal mass
- a palpable gallbladder – Courvoisier’s law states that the presence of a non-tender enlarged gallbladder means that jaundice is unlikely to be due to gallstones, as chronic cholecystitis causes the gallbladder to become shrunken and fibrotic. A palpable gallbladder is, therefore, a very worrying sign!
- nodular hepatomegaly
- ascites
- there may be cervical lymphadenopathy
- always check the supraclavicular lymph node (Virchow’s node) in the left side of the neck
Differential diagnosis – obstructive jaundice
Obstructive (or post-hepatic) jaundice can usually be distinguished from pre-hepatic or hepatic causes of jaundice by taking a history and doing some simple tests. The patient will have dark urine and pale stools, and may also have very itchy skin due to the deposition of bile salts. Alongside a raised conjugated bilirubin level, LFTs will show a cholestatic picture with an increase predominantly in ALP and GGT, with a less markedly raised ALT and AST. Cholesterol levels are also often raised.
The differential diagnosis for obstructive jaundice includes:
- intraluminal obstruction by:
- gallstones: stone in the common bile duct
- infection: liver flukes e.g. Clonorchis Sinensis
- drug-induced cholestasis: antibiotics, phenothiazines, corticosteroids, OCP/HRT
- mural thickening due to:
- biliary stricture: these can be congenital, or acquired as a result of gallstones, cholangitis or invasive procedures such as ERCP
- inflammation: primary biliary cirrhosis (PBC), primary sclerosing cholangitis (PSC)
- malignancy: cholangiocarcinoma
- extrinsic compression by:
- gallstones: cystic duct stone compressing common hepatic duct (Mirizzi’s syndrome)
- inflammation: pancreatitis, pancreatic pseudocyst
- malignancy: pancreatic cancer, gallbladder cancer, hepatocellular carcinoma, liver metastases, portal lymph node metastases, large gastric or duodenal cancer
Investigations
Diagnostic tests for patients with suspected pancreatic cancer include:
- history and clinical examination
- blood tests including FBC (for anaemia, white cell count and thrombocytosis), U+E (for renal function), LFTs (in obstructive jaundice these show raised bilirubin, ALP and GGT; a disproportionately raised ALT may be due to metastases), coagulation studies (for coagulopathy secondary to jaundice), glucose (for diabetes) and lipid profile (for disorders of cholesterol metabolism)
- if an endocrine tumour is suspected, additional blood tests include a fasting gut hormone screen
- tumour markers can be useful in making the diagnosis and monitoring response to treatment. Cancer antigen 19-9 (CA19-9) is a mucin glycoprotein antigen that is normally produced by the epithelial cells of the pancreas, gallbladder, bile ducts, GI tract, endometrium and prostate gland. It is overproduced in 75-85% of pancreatic cancer patients but may be normal in small, early-stage tumours, and 5% of the population are genetically unable to produce the antigen. The normal level is <37 units/ml; a level of >100 is suggestive of pancreatic cancer, and dramatically raised levels of >1000 are strongly associated with advanced unresectable disease. It is important to remember that a raised CA19-9 is not particularly specific for pancreatic cancer – it can also be raised in other gastrointestinal malignancies, pancreatitis, liver cirrhosis and inflammatory bowel disease. Carcinoembryonic antigen (CEA) levels may also be raised in up to half of pancreatic cancers, but this is not considered useful in making the diagnosis.
- transabdominal ultrasound can be used to assess for any obvious pancreatic tumours, as well as showing associated biliary dilatation, liver metastases, enlarged lymph nodes or ascites. It can also show other causes of jaundice such as gallstones. However, it is only able to accurately detect pancreatic cancer in 70-80% of cases, as the pancreas is a deep and elusive organ that often tries to hide behind gas-filled bowel loops or intraperitoneal fat.
- abdominal CT scan can provide a more detailed analysis of the pancreas and other abdominal organs and is much more accurate than ultrasound in detecting pancreatic cancer, with a sensitivity of about 90%. It can still miss some small tumours, especially those <3cm. It can also be used for CT-guided percutaneous biopsy, but this should be avoided in potentially resectable tumours as there is a risk of seeding cancer cells within the peritoneal cavity.
- endoscopic ultrasound (EUS) is a specialist investigation performed using a high-frequency ultrasound probe attached to an endoscope. This is positioned in the stomach or duodenum and used to image the pancreas from the inside. In experienced hands it is the gold standard diagnostic test for pancreatic cancer, with sensitivity and specificity of >99%. It can identify small tumours not visible on CT, and can also be used to assess resectability and guide fine-needle aspiration (FNA) sampling of the tumour for cytology to confirm the diagnosis. As well as being more accurate, EUS + FNA is also safer than percutaneous biopsy as the risk of peritoneal seeding is much lower.
- magnetic resonance cholangiopancreatography (MRCP) is often used in patients with obstructive jaundice who have had a non-diagnostic ultrasound scan. It is very useful in imaging the biliary tree and identifying causes of biliary obstruction such as CBD stones, strictures or cholangiocarcinoma. It is also useful in assessing cystic tumours. However, it is considered less accurate than CT in making the diagnosis of pancreatic cancer, and is also much more expensive and time-consuming.
- endoscopic retrograde cholangiopancreatography (ERCP) is an invasive procedure which is 90% accurate in identifying pancreatic cancers and can be particularly useful in assessing ampullary masses and unusual tumours such as IPMNs. It can be used to obtain brushings for cytology or biopsies for histopathological diagnosis, and can also allow therapeutic measures such as biliary stenting to be performed to relieve jaundice. However, there is a 5-10% risk of serious complications – including bleeding, sepsis, pancreatitis and perforation – and a 1% mortality associated with ERCP.
- laparoscopic ultrasound is an evolving alternative to endoscopic approaches
Staging investigations for patients with a confirmed pancreatic cancer include:
- triple-phase “pancreas protocol” CT scan is the gold standard for assessment of pancreatic cancer. By taking images at different times after IV contrast administration, it provides detailed imaging of the tumour itself, its invasion into surrounding tissues, the degree of vascular infiltration and biliary tree dilatation, and presence of lymphatic or liver metastasis. It can be used to accurately predict surgical resectability in up to 90% of cases.
- a bone scan or PET scan can be used to more accurately detect distant metastases
- diagnostic laparoscopy can also be used to rule out intraperitoneal spread in high-risk patients
- as discussed above, biopsies for histopathology can be obtained percutaneously, endoscopically via EUS/ERCP, laparoscopically or in some cases through an open procedure – endoscopic biopsy is felt to be the safest and most appropriate approach
Once all the required investigations have been performed, patients with pancreatic cancer should be referred to a specialist HPB cancer MDT where their case will be discussed on an individual basis by a team of surgeons, oncologists, radiologists and pathologists who make recommendations about the most appropriate course of treatment.
Grading – how aggressive is it?
The grading system for pancreatic cancer is based on several measures of the degree of differentiation within the tumour cells:
- Grade I – well-differentiated with plenty of mucin production and ≤5 mitotic figures (good)
- Grade II – moderately differentiated with irregular mucin production and 6-10 mitotic figures (OK)
- Grade III – poorly differentiated with minimal mucin production and ≥10 mitotic figures (bad)
Studies have shown that the grade of a pancreatic cancer is a useful prognostic indicator, in that patients with high-grade tumours tend to do much worse than their low-grade counterparts.
Staging – how far has it spread?
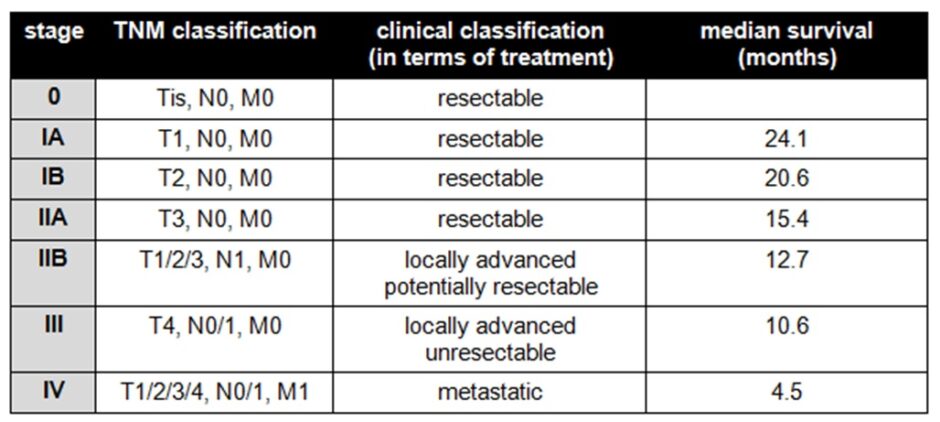
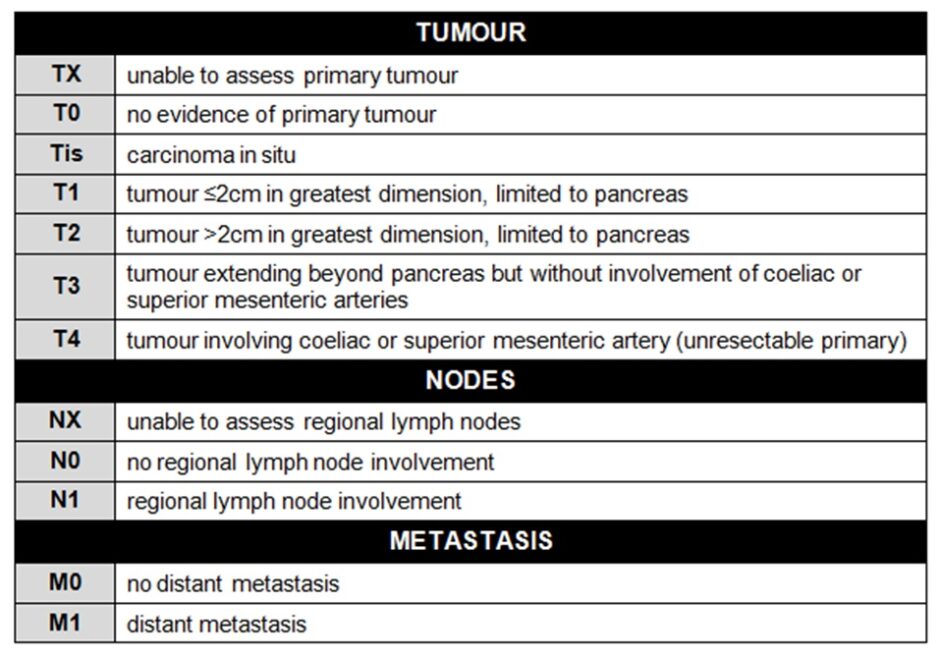
Pancreatic cancer is staged using the tumour nodes metastasis (TNM) system. Accurate staging is pivotal in informing prognosis and deciding the possible treatment strategies for pancreatic cancer, particularly in terms of whether a tumour is surgically resectable or not. Sadly, almost half of cancers are stage IV at diagnosis. The TNM criteria and their associated numerical staging stratification are shown in the tables below:
Management
Surgery is the only possible cure for pancreatic cancer, and watchful waiting is pretty much never an appropriate option. The management strategy is therefore primarily dictated by whether a tumour is resectable or not, but other important factors include the patient’s overall health and their fitness for major surgery and post-operative chemotherapy. There are surgical treatments available to both cure resectable cancers and palliate unresectable disease, and these are supported by the use of adjuvant medical therapies such as chemoradiotherapy and pain control measures.
Surgical management: curative
About 15-20% of ductal adenocarcinomas and 50-75% of other pancreatic cancers are suitable for active surgical treatment to remove the tumour. Surgical procedures to the pancreas carry very high risks, therefore it is vital that the patient is fit enough for the operation and that there is no evidence of metastasis on staging investigations or at the time of surgery in order to maximise the chances of long-term success. There are several surgical options available for pancreatic cancer, and the procedure performed depends on the location of the tumour.
Pylorus-preserving pancreaticoduodenectomy (“the modified Whipple”)
Patients with localised tumours in the head of the pancreas or periampullary region are suitable for pylorus-preserving pancreaticoduodenectomy, more famously known as the modified Whipple procedure. This is officially one of the most complex, difficult and daring operations known to surgical practice. It involves removal of the head of the pancreas, most of the duodenum, the common bile duct and the gallbladder, with or without extended dissection of additional lymph nodes. The traditional Whipple’s procedure also includes removal of the pylorus and antrum of the stomach, which is still occasionally necessary depending on the extent of the tumour. If the portal or superior mesenteric veins are involved, they can also sometimes be resected and reconstructed using vein grafts. Once all the above organs have been resected, the functional anatomy of the upper GI tract needs to be restored so the patient can still digest and absorb things. This is done by creating a Roux-en-Y loop from a segment of jejunum, and using end-to-side anastamoses to make:
- a pancreaticojejunostomy to drain pancreatic juice into the small bowel
- a hepaticojejunostomy to drain bile into the small bowel
- a duodenojejunostomy to restore GI tract continuity
This is normally done via a large abdominal “rooftop” incision, but some centres are starting to do it laparoscopically now. The Whipple is a high-risk procedure requiring careful pre-operative work-up and intensive post-operative monitoring and management. If performed at a specialist centre, the overall operative mortality rate is <5%.
Complications include bleeding, sepsis, bile and pancreatic leakage, anastamotic failure, delayed gastric emptying, nutritional problems and patients often end up in hospital for weeks or months while they recover. Patients require long-term pancreatic enzyme replacement in the form of Creon tablets taken with food. Five-year survival for patients with ductal carcinomas is 10-20% (the figures are low due to the fact that some surgeons use the Whipple as a palliative procedure too), and for patients with other types of pancreatic cancer is 40%.
For the super-keen amongst you, here is a nice video from tzanox MD on YouTube showing the key steps in a modified Whipple procedure. It’s a bit long, but I think they’ve done a great job condensing what was probably a 5-hour operation into 20 minutes. As you can see, it’s hardcore surgery involving a lot of messing about near massive scary blood vessels, and the anatomy is a lot more difficult to figure out in real life than it is on diagrams!
Distal pancreatectomy
Patients with localised tumours in the body or tail of the pancreas are suitable for a distal pancreatectomy. This involves removal of the body and tail of the pancreas as well as the spleen. This is technically a much more straightforward procedure than the Whipple and can be done openly or laparoscopically. Overall operative mortality is ~3%.
Complications are less frequent (as fewer organs have been chopped up and joined back together) but include bleeding, sepsis, pancreatic leakage and pancreatic endocrine insufficiency resulting in diabetes. Also, as they have had a splenectomy, these patients are at lifelong risk of potentially fatal overwhelming post-splenectomy infections (OPSI), usually due to encapsulated bacteria such as Streptococcus pneumoniae, Haemophilus influenza and Neisseria meningitidis. They, therefore, require pneumococcal, meningococcal and flu vaccinations as well as lifelong prophylactic antibiotics such as phenoxymethylpenicillin or erythromycin. Lots of patients wear alert bracelets to make medical staff aware they don’t have a spleen. If you ever see a patient who’s had a splenectomy, be vigilant for signs of sepsis, as it can progress very rapidly and can affect any asplenic patient regardless of their age, fitness or the time since their surgery.
Total pancreatectomy
Rarely, patients with localised tumours in or involving the neck of the pancreas, or diffuse cancers such as IPMN, may be offered a total pancreatectomy. This combines both of the procedures described above and is clearly a major undertaking, which is reflected in its operative mortality of 8-10%. Complication rates are similar to a Whipple procedure but with the added bonus of the risk of post-splenectomy sepsis and guaranteed lifelong insulin-dependent diabetes, which can be very brittle and difficult to manage.
Surgical management: palliative
Patients presenting with obstructive symptoms secondary to inoperable pancreatic cancer may be offered palliative surgical treatments to help control their symptoms:
- biliary obstruction and jaundice is a very common problem in advanced disease. It leads to symptoms of malaise, abdominal pain and severe pruritis, and also puts patients at risk of death from cholangitis, renal failure and severe coagulopathy. Options for management include biliary stenting, either endoscopically via an ERCP or percutaneously using radiological transhepatic techniques, and biliary bypass surgery with choledochojejunostomy, hepaticojejunostomy or sometimes even a palliative Whipple procedure.
- duodenal obstruction is reasonably common and presents with symptoms of gastric outlet obstruction such as epigastric pain and profuse non-bilious vomiting. It can be treated endoscopically with duodenal stenting, or with gastric bypass surgery using a gastrojejunostomy or Roux-en-Y bypass.
- pancreatic duct obstruction can lead to pain and malabsorption. It can be treated with endoscopic pancreatic stenting or sometimes with pancreaticoduodenectomy.
Pain is a significant problem in patients with pancreatic cancer. This is generally managed with opiate analgesia, neuropathic adjuncts and management of obstructive symptoms as described above, but this is not always enough. Advanced pain control measures include alcohol ablation of coeliac plexus nerves, which can be performed transthoracically, transabdominally or transgastrically under EUS guidance.
Newer minimally invasive treatments for inoperable pancreatic cancer include irreversible electroporation (IRE), also known as NanoKnife. This is an innovative non-thermal cell destruction technique which uses the direct application of short bursts of high-voltage electric current to destroy cancer cells. Because it does not generate high temperatures, it minimises damage to surrounding tissues such as blood vessels and bile ducts. It has been shown in American trials to be an effective treatment with the potential to double survival, but can cause lethal cardiac arrhythmias if the electric current is not properly synchronised with the patient’s cardiac cycle. It is currently only approved by NICE for use in clinical trials, but may be used to treat a variety of cancers in the future.
Medical therapies
Medical therapies for exocrine cancers may be used as adjuncts to surgery, or as a primary palliative treatment in advanced disease:
- patients undergoing curative surgical treatment of their cancer should receive a gemcitabine-based adjuvant chemotherapy regime post-operatively. This was shown by the ESPAC-1 trial to double five-year survival from 10% to 20%. The ESPAC-4 trial is currently investigating combination chemotherapy regimes to see if this can be improved further. Adjuvant radiotherapy is not used, as it does not improve survival and may actually be harmful.
- patients with unresectable disease may be offered palliative chemotherapy – although most pancreatic tumours are fairly chemoresistant, gemcitabine-based regimes can delay disease progression and improve survival in patients with reasonable performance status.
- Radiotherapy can help with pain control but does not improve survival.
- Pancreatic enzyme supplementation with Creon tablets can help with malabsorption and weight loss.
- advanced metastatic cancer (which is unfortunately very common) requires individualised palliative treatment with chemoradiotherapy and symptom control.
Unresectable or metastatic functional endocrine tumours (apart from somatostatinomas) can be managed medically with somatostatin analogues such as octreotide and lanreotide – these act to control symptoms by suppressing pancreatic hormone secretion.
Follow-up and prognosis
- the overall prognosis for pancreatic cancer is abysmal (20% 1-year survival, 3.7% 5-year survival, and just 1% 10-year survival)
- mortality rates have not really improved since the 1970s, despite heroic efforts by armies of researchers, surgeons and oncologists around the world
- even in the minority of patients who undergo surgery with a curative intent, median survival is only 11-20 months, with a 5-year survival of 7-25%
- for patients with locally advanced unresectable tumours, there is a median prognosis of 6-11 months from the time of diagnosis
- patients with metastatic disease have a tragically short median prognosis of 2-6 months from diagnosis and tend to decline very rapidly
- survival varies depending on the type, grade and stage of cancer – the small subgroup of patients with pancreatic endocrine tumours tend to have a much better prognosis
- patients who receive surgical treatment for their cancer are usually followed up by a specialist HPB (hepatopancreatobiliary) surgical unit with CA19-9 monitoring and regular CT scans
- patients with advanced incurable disease may be referred for management by oncology or palliative care specialists, either as inpatients or in the community
Prevention strategies
There are a number of public health interventions in place which aim to reduce the incidence of pancreatic cancer, as well as a range of other lifestyle-related illnesses. Examples would include education and awareness campaigns which encourage smoking cessation, a healthy alcohol intake and the reduction of lifestyle risk factors such as obesity and poor diet.
It is clear that the development of a population screening programme to catch pancreatic cancer at an early treatable stage would be extremely valuable, as it is a disease that characteristically presents at a late and untreatable stage. Unfortunately, at the moment there is no available test that is sufficiently accurate, safe and straightforward to justify screening the population as a whole. CA19-9 is simply not sensitive or specific enough. A study called the EUROPAC trial is currently underway to test a screening programme for high-risk patients with a significant family history, such as those with hereditary pancreatitis, familial pancreatic cancer or cancer syndromes such as BRCA2. The screening protocols being tested involve either a CT scan or EUS every 3 years from the age of 40.
References
Images:
- Blausen.com staff (2014). “Medical gallery of Blausen Medical 2014”. WikiJournal of Medicine 1 (2). DOI:10.15347/wjm/2014.010. ISSN 2002-4436. [CC BY 3.0], from Wikimedia Commons
- By James Heilman, MD [CC BY 3.0], from Wikimedia Commons
Further reading:
- Adkisson CD, Harris AS, Bridges MD et al; “Solid pseudopapillary tumor of the pancreas: Report of five cases“. International Journal of Hepatobiliary and Pancreatic Diseases 2012;2:9-14
- Cancer Research UK – Cancer incidence for common cancers -Available from: [LINK]
- Cancer Research UK – Cancer mortality for common cancers – Available from: [LINK]
- Cancer Research UK – Pancreatic cancer – Available from: [LINK]
- “Guidelines for the management of patients with pancreatic cancer periampullary and ampullary carcinomas“; Pancreatic Section of the British Society of Gastroenterology: Gut 2005;54:v1-v16
- Medscape eMedicine – Pancreatic cancer – Available from: [LINK]
- Patient UK – Pancreatic Exocrine Tumours -Available from: [LINK]
- Radiology Assistant – Pancreas cystic lesions – Available from: [LINK]
- Radiopaedia – Pancreatic ductal carcinoma – Avaiable from: [LINK]
Reviewer
Mr Avinash Sewpaul
ST8 in HPB & Transplant Surgery