- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
Parkinson’s disease (PD) is a neurodegenerative disorder occurring due to the loss of dopaminergic cells from the substantia nigra area of the brain.
Cardinal symptoms of PD are those of movement disorder, although non-motor symptoms such as cognitive impairment are also widely prevalent and account for a significant symptomatic burden. It is the second most common neurodegenerative disorder.1
Parkinson’s disease is the most common cause of parkinsonism, a syndrome of motor symptoms that involves bradykinesia plus at least one of; rigidity, tremor or postural instability.2
Aetiology
PD typically presents in those above 55 years old. It has a prevalence of 1-2% in people over 65 and this rises with age.3 There is a higher prevalence of PD in men, with a male to female ratio of 1.5:1.1
Pathophysiology
Dopamine is a key neurotransmitter used in the regulation of movement. It is produced in the substantia nigra within the basal ganglia which is the key movement control centre of the brain (Figure 1).
In PD, there is a progressive loss of dopamine-producing (dopaminergic) neurons. There are several dopaminergic pathways affected, but the pathognomonic motor signs of Parkinson’s disease occur due to the loss of dopaminergic neurons in the nigrostriatal pathway, a motor control pathway that originates in the substantia nigra and terminates in the striatum.
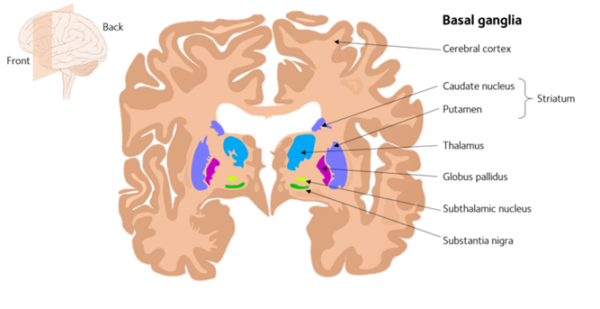
Physiologically, dopamine (an excitatory neurotransmitter) acts to upregulate the thalamus and ultimately the motor cortex via the direct pathway of the basal ganglia. This results in increased movement.
Loss of these dopaminergic neurons results in a reduction in the action of the direct pathway of the basal ganglia and a resultant increase in the antagonistic indirect pathway, which has a restrictive action on movement. Bradykinesia and rigidity are therefore key features of parkinsonism.
In PD, there is also formation of protein clumps (Lewy bodies) within the remaining neurons. These typically begin to form in the lower structures of the brainstem and progressively spread cranially towards the cortex. This progression can be broken down into Braak stages, however, this is a post-mortem staging progress and is not used clinically.
Risk factors
PD is an idiopathic disease. It is not known what causes the loss of dopaminergic cells, although there appears to be some genetic associations, with a first-degree family history cited in up to 20% of cases.3
Male sex and increasing age are known risk factors for PD. Research is ongoing into environmental factors and triggers of PD. Smoking is thought to be protective.
Clinical features
Parkinson’s disease is usually a clinical diagnosis, based on history and examination findings. The disease may remain sub-clinical for a prolonged period, with many features not clinically apparent until at least 50% of dopaminergic cells have been lost.3
History
Typical symptoms of Parkinson’s disease include:
- Motor symptoms: slowness of movement, stiffness, tremor, instability or falls, reduced dexterity
- Autonomic symptoms: constipation, light-headedness on standing, excessive salivation or sweating, urinary dysfunction, sexual dysfunction
- Mood/cognitive symptoms: low mood, anxiety, apathy
- Sleep disturbance and daytime sleepiness
- Anosmia or reduced sense of smell
Other symptoms which may be reported by family or carers include:
- Cognitive decline
- REM sleep behaviour disorder: manifests with behaviours such as punching, kicking or shouting during sleep
- Altered handwriting: micrographia
Although motor symptoms are the diagnostic features of parkinsonism, non-motor features may precede motor symptoms by years in PD.
Other important areas to cover in the history include:
- Past medical history: history of known tremor or movement disorder, cognitive impairment, frequent falls
- Drug history: drug-induced parkinsonism must be excluded (see differential diagnosis section)
- Family history: first-degree relatives with PD diagnosis
- Social history: smoking is thought to be protective for PD and alcohol withdrawal is a common cause of tremor. It is important to ask about the patient’s living situation, mobility, carers and activities of daily living.
Clinical examination
A thorough neurological exam should be carried out on all patients suspected of Parkinson’s disease. For more information, see the Geeky Medics OSCE guide to Parkinson’s disease examination.
Typical clinical findings on neurological examination include:
- Bradykinesia: Tested using finger taps which are slow with progressive fatiguing and decrease in amplitude
- Tremor: typically a ‘pill-rolling’ tremor of the hands at rest
- Rigidity
- Abnormal gait: shuffling, festinating (progressive acceleration of stride alongside a reduction of stride length), reduced arm swing, freezing (particularly when turning or when faced with doorways, obstacles or changes in terrain)
- Postural instability: tested using the pull test, although not usually present at diagnosis
Motor features usually begin unilaterally but will progress to become bilateral. The side affected first continues to be worse causing persistent asymmetry.
Other clinical findings on general examination may include:
- Hypomimia: decreased facial expression
- Postural hypotension
- Confusion
- Reduced dexterity: struggling with functional tasks such as fastening buttons
Differential diagnoses
Parkinsonism
Other causes of parkinsonism include:3,5
- Lewy body dementia
- Multiple system atrophy: (MSA) unsteadiness, falls and autonomic dysfunction may be more predominant features of early disease in MSA
- Progressive supranuclear palsy (PSP): unsteadiness, dysphagia and gaze palsies may be more predominant features of early disease in PSP
- Vascular parkinsonism: patients may report a history of cerebrovascular disease along with a sudden onset of symptoms
- Drug-induced parkinsonism
Drug-induced parkinsonism
Any drug which acts by blocking dopamine or increasing its breakdown can cause parkinsonism.
Common causative drugs include:
- Antipsychotics: first-generation antipsychotics such as haloperidol are more likely to have parkinsonian side effects than second-generation agents
- Anti-emetics: prochlorperazine, metoclopramide
Symptoms are more likely to be bilateral in drug-induced parkinsonism than in PD. Culprit drugs should be reduced, replaced or stopped if clinically safe to do so to reduce parkinsonian symptoms.
Essential tremor
Essential tremors are common. They can occur at any age and patients will often have a family history of essential tremor. Like drug-induced parkinsonism, essential tremor is usually bilateral.
The tremor in PD is typically noted in the hands, however, essential tremor is often present in the head, neck and trunk as well as the hands.
Essential tremor increases on action and improves at rest, the opposite of PD. It also improves with alcohol or beta-blockers but may be worse after caffeine or in times of heightened stress.3
Investigations
Bedside investigations
Relevant bedside investigations include:
- Lying and standing blood pressure: patients with PD may have postural hypotension
- Cognitive assessment (e.g. Montreal cognitive assessment or abbreviated mental test)
Laboratory investigations
There is no routine testing for biochemical markers of Parkinson’s disease.
In the case of cognitive impairment, a ‘confusion screen’ set of tests should be considered, to exclude reversible causes of confusion/delirium.
Relevant laboratory investigations as part of a confusion screen include:
- Full blood count: low haemoglobin or raised white cell count may indicate anaemia or infection respectively
- Urea & electrolytes: electrolyte derangement
- Liver function tests: Hepatic encephalopathy
- Inflammatory markers (CRP): infection/inflammation
- Glucose and HbA1c: hypo/hyperglycaemia
- Thyroid function tests: hypothyroidism
- Calcium: hyper/hypocalcaemia
- B12, folate and haematinics: anaemia
Imaging
Dopamine active transporter (DaT) scans can be used. These scans measure dopamine uptake in the basal ganglia and are therefore specific for primary Parkinsonian syndromes (i.e. not drug-induced parkinsonism or essential tremor).
Unfortunately, they are very costly and therefore reserved only for cases of diagnostic uncertainty.
Other investigations
Post-mortem findings include a reduction in the volume of the substantia nigra and the formation of Lewy bodies.
Diagnosis
A movement disorder/PD specialist should make the formal diagnosis of PD. Patients with suspected PD should be referred promptly (and untreated) to a specialist clinic.6
Key diagnostic features are bradykinesia plus one of the following symptoms:2
- Rigidity
- Tremor
- Postural instability
Management
There is no cure for PD and current treatment does not halt or reverse the progression of the disease.
Medical management
Medical management should be initiated by a PD specialist.
Treatment aims to reduce symptoms by replacing dopamine or reducing dopamine breakdown. This can happen in a variety of ways:6
- Levodopa: often first-line treatment. Levodopa is a precursor of dopamine and is therefore metabolised into dopamine. It is taken in conjunction with carbidopa (a dopamine decarboxylase inhibitor) to reduce metabolism into dopamine before the drug crosses the blood-brain barrier. This reduces side effects such as nausea and vomiting.
- Monoamine oxidase-B inhibitors (MOA-B inhibitors): these drugs block the breakdown of dopamine (e.g. selegiline).
- Dopamine agonists: these drugs bind to and stimulate dopamine receptors and therefore mimic the action of dopamine (e.g. ropinirole and rotigotine patches). A major potential side effect which patients must be counselled about is impulse control disorders. This is because alternative dopamine pathways in the limbic system are also agonised which can alter attitude to risk.
- Catechol-O-methyl transferase (COMT) inhibitors: these drugs decrease dopamine breakdown and prolong the action of levodopa (e.g. entacapone). COMT inhibitors are always taken alongside levodopa.
Anti-parkinsonian medications are ‘critical medications’ and are time-sensitive. Patients who miss doses or receive late doses could experience freezing (akinesia). Suddenly stopping PD meds may also precipitate neuroleptic malignant syndrome which can be life-threatening.3
Response to medication deteriorates with time and side effects may be seen such as dyskinesias (typically seen soon after taking medications due to overstimulation) or re-emergence of symptoms as each dose wears off. With disease progression and increased loss of dopaminergic cells, increasing doses of medication are needed, leading to an increased risk of side effects.
Multidisciplinary management
Multi-disciplinary input is a key part of the holistic bio-psycho-social management of PD. Professionals involved should include:
- Physiotherapists
- Speech and language therapists (SALT)
- Parkinson’s nurse specialist
- Occupational therapists
Deep brain stimulation
Deep brain stimulation is a specialised management option and not often used. It is reserved for those with severe symptoms refractory to medical management yet still fit with few co-morbidities.6
Complications
Poor mobility can lead to increased falls and resultant injuries. Reduced independence is also seen as the disease progresses and mobility deteriorates.
The risk of dementia is significantly higher in PD than in the general population, namely dementia with Lewy bodies (DLB) and Parkinson’s dementia. These syndromes present clinically and pathologically very similarly, however, the clinical timeline distinguishes them:
- DLB: cognitive deterioration presents before or alongside parkinsonian motor symptoms
- Parkinson’s dementia: movement disorder symptoms preceded cognitive decline by a year or more
Hallucinations are a common complication of PD. Insight is generally good, however, if this is lost, patients can develop paranoia.
Aspiration pneumonia is frequently seen in end-stage PD as a complication of dysphagia. This increases morbidity and mortality.
Key points
- Parkinson’s disease is an idiopathic progressive neurological disorder. It is the most common cause of parkinsonism.
- Risk increases with age and PD is more prevalent in men than women.
- Pathophysiology involves the loss of dopaminergic neurons from the substantia nigra area of the basal ganglia resulting in the dysfunction of neural pathways key to movement.
- Diagnosis is clinical with classical motor symptoms of bradykinesia, rigidity, tremor and postural instability.
- Non-motor symptoms include sleep disturbance, autonomic dysfunction and cognitive decline with an increased risk of dementia (specifically DLB and Parkinson’s dementia).
- Symptoms are progressive and there is currently no cure.
- Management should be specialist-led and is aimed at increasing the activity of dopaminergic pathways.
- Complications include cognitive decline and autonomic disorders, both of which increase the risk of falls.
Reviewer
Professor Richard Walker
Consultant geriatrician
Editor
Dr Chris Jefferies
References
- Patient.info. Parkinsonism and Parkinson’s Disease. Published in 2018. Available from: [LINK]
- Hughes, Daniel, Kilford, Lees. UK Parkinson’s disease society brain bank clinical diagnostic criteria. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. Published in 1992. Available from: [LINK]
- NICE Clinical Knowledge Summary. Parkinson’s disease. Published in 2022. Available from: [LINK]
- Beckie Port, adapted from original work by Jlienard, previously derived from work by Andrew Gillies’, Mikael Häggström’s and Patrick J. Lynch’s. The structures of the basal ganglia. Licence: [CC BY-SA]
- Parkinson’s UK. Types of parkinsonism. Published in 2019. Available from: [LINK]
- Parkinson’s disease in adults. NICE Guideline. Published in 2017. Available from: [LINK]




