- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
The coagulation cascade is a series of reactions, catalysed by protein enzymes known as coagulation ‘factors’. When activated, these factors trigger the conversion of other factors in the coagulation cascade resulting in secondary haemostasis.
Secondary haemostasis refers to the stabilisation of activated platelets due to the actions of strands of fibrin. These generate a ‘stable mesh’ the main component in a thrombus (clot).
The activation and adherence of platelets to the damaged endothelium (primary haemostasis), and vascular response to injury (e.g. vasoconstriction) are the other key components involved in thrombus formation and haemostasis.
Fibrin is converted from its precursor fibrinogen by thrombin and is the end-product of the chain of reactions that make up the coagulation cascade.
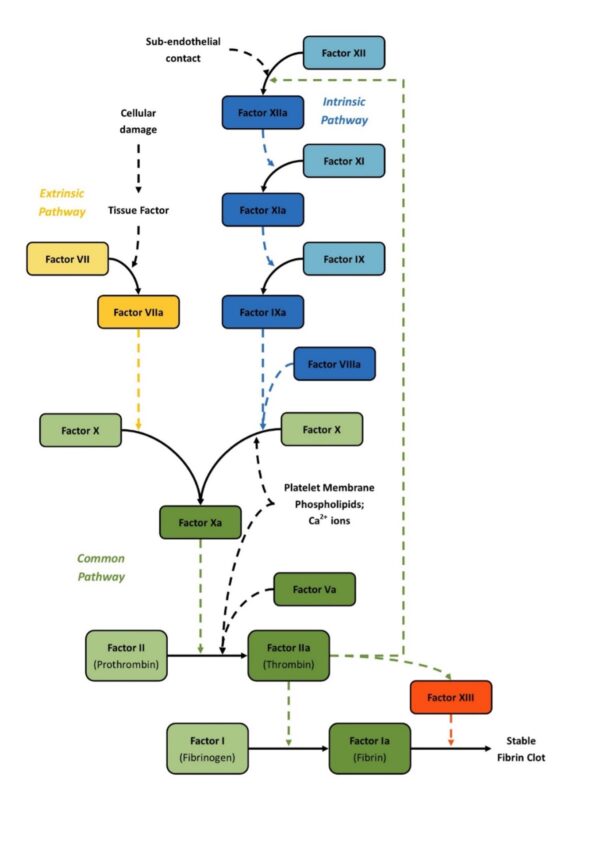
The coagulation cascade can be divided into three segments or pathways: the intrinsic, extrinsic and common pathways. Coagulation is more complex than its depiction in the coagulation cascade, but the cascade is a useful starting point to understanding laboratory tests of coagulation.
Coagulation factors
Coagulation factors are usually designated a number in Roman numerals with a letter ‘a’ after the numeral when in its active form. For example, activated factor X is referred to as factor Xa.
Some factors are more commonly known by their name rather than their number. For example, factor II is usually referred to as thrombin.
The extrinsic pathway
Initiation of the coagulation cascade is usually mediated by tissue factor via the extrinsic pathway.
Tissue factor is expressed on the surface of many cells found outside blood vessels but not on the surface of circulating blood cells or the endothelium. When the endothelium is damaged, tissue factor comes into contact with blood and combines with circulating factor VII to form a complex that leads to the activation of factor X, triggering the common pathway.
The common pathway
The common pathway begins with activation of factor X (to factor Xa) via either the extrinsic pathway or the intrinsic pathway. It is the final stage of the coagulation cascade and leads to the formation of thrombin and fibrin.
Factor Xa combines with factor V, platelet membrane phospholipids and Ca2+ ions to convert prothrombin into thrombin. Thrombin then converts fibrinogen into fibrin strands which form an important structural component of a thrombus.
In addition to the formation of fibrin, thrombin activates many parts of the coagulation cascade via a positive feedback loop through the intrinsic pathway leading to the formation of large amounts of additional thrombin (a ‘thrombin burst’).
The intrinsic pathway
The intrinsic pathway can be activated by surface contact, when damage to the vascular endothelium leads to exposure of clotting factors to negatively charged subendothelial surfaces, mediated by the molecule Kallikrein.
Within blood vessels, thrombin generated from previous activation of the extrinsic pathway also activates the intrinsic pathway.
The series of reactions in the intrinsic pathway leads to initiation of the common pathway and therefore amplification of thrombin production through the creation of a positive feedback loop.
Reactions in the intrinsic pathway include:
- Surface contact activates factor XII
- Factor XIIa activates factor XI
- Factor XIa activates factor IX
- Factor IXa combines with factor VIIIa, platelet membrane phospholipid and Ca2+ ions to activate factor X
- Factor Xa activates the common pathway generating more thrombin
This continuous cycle means that vast amounts of thrombin can be generated from a single initial stimulus and a thrombus can be formed quickly after injury to limit blood loss.
Actions of thrombin
Thrombin plays a significant role in many aspects of coagulation and haemostasis:
- Fibrin generation: thrombin converts fibrinogen into fibrin
- Intrinsic pathway activation: thrombin generates a positive feedback loop by initiating the intrinsic pathway
- Factor XIII activation: thrombin converts factor XIII into factor XIIIa (fibrin stabilising factor) which then cross-links fibrin to generate a ‘fibrin mesh’ which encapsulates activated platelets creating a thrombus and stopping bleeding
- Platelet activation: thrombin receptors on platelets cause activation and aggregation, further enhancing the haemostatic effects of the coagulation cascade
- Regulation of clot formation
An overview of the coagulation cascade
Table 1. An overview of the coagulation cascade.
| Pathway | Activation | Components | Outcome |
|
Extrinsic |
Tissue factor: released due to vascular damage |
|
Activation of factor X |
|
Intrinsic |
Surface contact: exposure of coagulation factors to negatively charged vascular subendothelial surfaces Thrombin: released at the end of the common pathway (positive feedback loop) |
|
Activation of factor X |
|
Common |
Factor Xa: activation of factor X by either the extrinsic or intrinsic pathway |
|
Generation of thrombin:
|
Regulating the coagulation cascade
Without regulatory mechanisms, the positive feedback loop triggered by thrombin in the intrinsic pathway would continue indefinitely and cause dangerous levels of clotting in the blood.
For this reason, various proteins inhibit aspects of the coagulation cascade once enough thrombin has been produced.
Protein C is produced in response to thrombin binding to the receptor thrombomodulin on the vascular endothelium. This leads to protein S activation which then breaks down factors Va and VIIIa in a negative feedback loop.
Antithrombin is a natural anticoagulant that is produced by the liver and destroys factors XIa, Xa and thrombin.
Heparin and fondaparinux enhance the natural effects of antithrombin in inhibiting thrombin and factor Xa.
Tissue factor pathway inhibitor is a protein that binds to and inactivates factor VIIa and factor Xa.
Vitamin K and Warfarin
Warfarin is a vitamin K antagonist which prevents the synthesis of vitamin K-dependent clotting factors (II, VII, IX and X).
Low molecular weight heparin (LMWH) is prescribed alongside warfarin when treatment is initiated. This is because vitamin K is also required for the synthesis of the natural anticoagulants protein C and protein S so Warfarin may inhibit their production initially. LMWH can be stopped once the INR is at the target range.
Coagulation screening
Multiple tests exist to assess haemostasis. They involve adding triggers to a sample of reversibly anticoagulated plasma (by citrate) and measuring the time taken for fibrin to form.
The two most frequently performed tests are the prothrombin time (PT) and the activated partial thromboplastin time (APTT).
For more information on interpreting these tests, see the Geeky Medics guide to interpreting a coagulation screen.
Prothrombin time (PT)
The prothrombin time measures the time taken for fibrin to form via the extrinsic pathway.
The normal range is 9-12 seconds. Factors measured include factor VII; factor X; factor V; prothrombin and fibrinogen.
Causes of a prolonged PT include:
- Disseminated intravascular coagulation (DIC)
- Vitamin K deficiency (Factor VII is the vitamin K dependent clotting factor with the shortest half-life)
- Chronic liver disease
Prothrombin time is the test that forms the basis of the INR (international normalised ratio), a ratio of the patient’s PT compared to a normal PT.
The INR is used to monitor the levels of anticoagulation in patients on Warfarin and adjust the dosage as necessary.
Activated thromboplastin time (APTT)
The activated thromboplastin time measures the time taken for fibrin to form via the intrinsic pathway.
The normal range is 23-38 seconds. Factors measured include factor XII, factor XI, factor IX, factor VIII, factor X, factor V, prothrombin and fibrinogen.
Causes of a prolonged APPT include:
- Disseminated intravascular coagulation (DIC)
- Clotting factor deficiencies (e.g. haemophilia A or B)
- Lupus anticoagulant
- Von Willebrand’s disease
Common pitfalls
Despite measuring the activation of each coagulation pathway, the processes that form clots in samples of anticoagulated blood do not always accurately reflect a patient’s ability to clot and therefore should be interpreted with caution, alongside the patient’s history and other investigations.
For example, the PT assesses the extrinsic pathway after it has been initiated by tissue factor but does not account for the fact that in vivo (within circulating blood) large amounts of thrombin are produced via a positive feedback loop through the intrinsic pathway, with even small amounts of factor VII and this alone can bring about haemostasis.
Another example is that the APTT can be interfered with by antibodies against phospholipids (Lupus anticoagulants) which prolong the APTT test but do not increase the tendency of a patient to bleed.
Dysfunction of the coagulation cascade
Dysfunction of the coagulation cascade can be caused by several medical conditions:
- Haemophilia is a congenital deficiency in either factor VIII (haemophilia A), factor IX (haemophilia B) or factor XI (haemophilia C) leading to defective clotting and therefore a tendency to bleed.
- Chronic liver disease can lead to defective coagulation as many coagulation factors are depleted as the liver’s synthetic function is impaired.
- Factor V Leiden is a common genetic mutation to the gene coding for Factor V. It increases the resistance of factor V to activated protein C making it more difficult to break down and therefore leading to an increased tendency to clot.
Key points
- The coagulation cascade is one of the key components in the cessation of bleeding (haemostasis), by generating a fibrin mesh that stabilises activated platelets
- Two pathways (the extrinsic and intrinsic) both result in the activation of factor X, which leads to the conversion of prothrombin into thrombin
- Thrombin converts fibrinogen into fibrin and triggers the activation of factor XIII to cross-link the fibrin strands and stabilise the clot
- A stable clot is formed by activated platelets, fibrin and factor XIIIa
- Dysfunction of the coagulation cascade (including factor deficiencies) is responsible for many of the most common bleeding and clotting disorders (e.g. haemophilia, factor V Leiden)
- Laboratory investigations (a coagulation screen) can be used to assess parts of the coagulation cascade, but not through the same processes that occur naturally, and should be interpreted with caution
Reviewer
Dr Angela Collins
Haematology Consultant
Editor
Dr Chris Jefferies
References
- Hoffbrand A. V., Moss P. A. H., 2011., Essential Haematology, 6th Edition, John Wiley & Sons, New York
- Ralston S. H., et al., 2018., Davidsons Principles and Practice of Medicine, 23rd Edition, Elsevier, Edinburgh
- Naish J., Syndercombe Court D., 2019., Medical Sciences, 3rd Edition, Elsevier, Edinburgh
- Hall J. E., Hall M. E., 2021., Guyton and Hall Textbook of Medical Physiology, 14th Edition, Elsevier, Edinburgh




