- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
A brief history of pacing
The concept of dying from arrhythmias was first considered by Hippocrates was who quoted as saying:
‘Those who die from frequent and strong faints without any manifest cause die suddenly’
This was refined over the years until Geronimo Mercuriale formulated the concept of syncope and related it to a slow pulse in the 1500s.
Electricity was first factored into the equation when a variety of physicians (Nickolev Abilgaard in 1775 and Alexander von Humboldt in 1797) applied electrodes to a variety of birds and probably performed the first cardioversions!
Catharina Serafin, an unskilled Prussian labourer, played a crucial role in the foundations of pacing. She had a left-sided chest wall tumour that had exposed her heart – Hugo von Ziemssen then used electrodes to stimulate and change her heart rate at will. What Catharina got out of the bargain is unclear…
Karl Wenkebach described the heart block that now bears his name in 1899 (before the ECG was invented – he traced the jugular venous pulsation and radial artery pulse simultaneously) and John Hay identified type II second degree AV block in 1906 by similar means. Woldemar Mobitz then stole the glory by matching these to ECG findings and the Mobitz criteria was born.
Pacemakers were first used in the 1950s and were large, bulky external devices run off mains electricity. Earl Bakken (co-founder of Medtronic) began the process of miniaturisation when has subsequently led to the implantable devices of today.
See this article, for more fascinating history.
Constituent parts of a pacemaker
Generator and leads
Generator
The generator is a physical box filled with electronics that allow the pacemaker to generate its impulses and function.
Leads
Leads come in two varieties: active or passive. Active leads come equipped with small screws which are used to secure them into the myocardium and increase stability. However, in older people, this increases the risk of perforation. passive leads do not have a screw on the end and are wedged into the trabeculae in the right ventricle (they are therefore more likely to displace).

Pacemaker implantation
Pacemakers are implanted under local anaesthetic in the subclavian region on either the left or the right side (depending on whether the patient is left or right-handed to avoid the dominant hand).
The leads are placed via the cephalic vein (located into the deltopectoral groove under direct vision) or the subclavian vein (located by blind puncture under the clavicle where it crosses the first rib with an associated risk of pneumothorax during implantation).
Incision location
The location of the incision and pre-pectoral pocket (made in the fascial plane between the subcutaneous fat and the pectoral muscle) depends on the approach being taken.
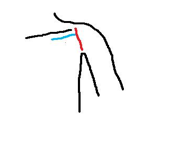
Blue line
This is the typical incision made for a planned subclavian approach parallel to the clavicle (drawn in black).
Red line
This is the typical incision for a planned cephalic approach in the deltopectoral groove.
Placing the leads in the heart
The right atrial lead is then passed to the right atrial appendage and the right ventricular lead is passed to the right ventricular apex or to a position on the ventricular septum (to allow pacing of the bundles of His mimicking normal conduction as closely as possible).
Left ventricular pacing leads (see cardiac resynchronization therapy section) are placed into the coronary sinus veins around the exterior of the heart to pace the left ventricle. Leads are tested before being sewn in and connected to the generator which is then implanted in the pre-pectoral pocket.
How does a pacemaker work?
Most pacemakers are demand pacemakers. This means that they only pace on demand (e.g. when the heart rate is less than 60). To achieve this, the pacemaker has two functions: pace or inhibit.
Let’s take a pacemaker with a single ventricular lead and run through a cardiac cycle:
- A ventricular contraction is sensed by the pacemaker. This begins the RR delay (in this case 1 second to correspond to 60 beats per minute).
- If another ventricular contraction is sensed before this interval has finished, the pacemaker senses this, inhibits any pacing and starts the clock again.
- If the interval then elapses without a further ventricular contraction, the pacemaker paces and starts the clock again.
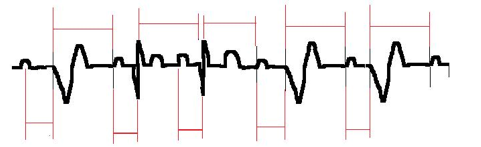
As you can see, this means that the pacing spikes on an ECG can be intermittent or absent if the patient’s heart rate is above the threshold for demand pacing. This does not mean the pacemaker is broken!
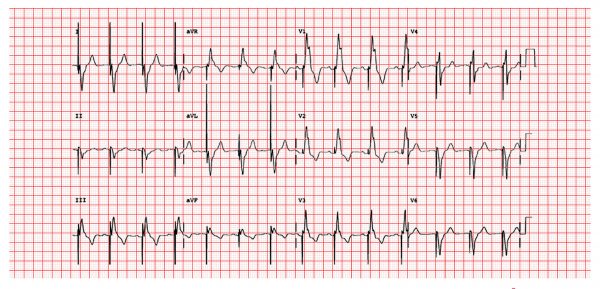
Dual-chamber pacemakers
Dual-chamber pacemakers (right atrial and ventricular leads) work using a similar system (although obviously slightly more complex) triggered initially by the P wave. The tracing below demonstrates all the combinations possible:
- The first intrinsic P wave is sensed by the atrial lead (A SENSED). This starts the PR delay – marked in red. If there is no intrinsic ventricular activity during this period, the pacemaker paces the ventricle (V PACING). This commences the RP delay.
- This elapses without intrinsic atrial activity so the pacemaker paces the atrium (A PACED). During the PR interval, the pacemaker senses ventricular activity and stops the generator sending an impulse (V SENSED). This commences the RP delay.
- The third complex is entirely intrinsic (A SENSED/ V SENSED). The pacemaker simply senses the intrinsic activity and allows things to go on as normal.
- The fourth and fifth complexes are entirely paced (A PACED/V PACED). There is no intrinsic heart activity before the intervals expire and the pacemaker steps in to take over.
How many leads do you need?
Pacemaker implants usually have one, two or three leads.
The atrial lead is placed in the right atrium to detect organised atrial contraction. If the patient is in permanent atrial fibrillation (AF), there is no organised atrial contraction, so an atrial lead is not required. These patients therefore only get a right ventricular lead.
Pacemakers can sense when patients with paroxysmal AF go back and forth and can change mode accordingly turning the A lead on and off (MODE SWITCHING). These patients intermittently have organised atrial contraction and as such benefit from both an atrial and ventricular lead.
Some patients with sinoatrial node disease but excellent AV node conduction (i.e. young patients) have an atrial pacemaker that only has an atrial lead. However, older patients who are more likely to have or develop an AV node block will benefit in the long term from a dual-chamber implant.
In summary:
- Atrial lead only → Sino-atrial disease in young people with good AV node conduction
- RV lead only → Pacing whilst in permanent atrial fibrillation
- Dual-lead → All other scenarios
Pacemaker lead appearance on a chest X-ray
The chest radiographs below show the typical appearance of pacemaker leads:
- The atrial lead is placed in the right atrial appendage and should usually be upwards pointing. The extra loop of lead allows the placement of the generator without pulling on the lead.
- The right ventricular lead is placed either in the RV apex (as seen here), or on the RV septum. The lead will then be pointing upwards towards the upper heart border.
- As the right heart chambers are anterior to the left heart chambers, the leads should both point anteriorly on the lateral film.
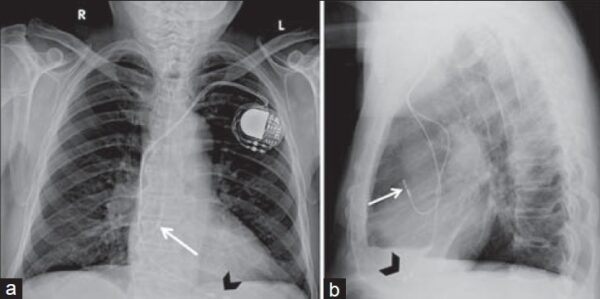
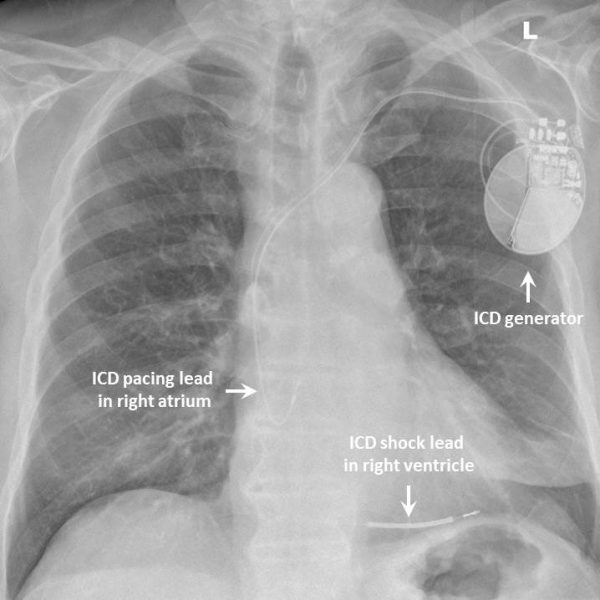
Implantable cardioverter-defibrillators (ICD)
An implantable cardioverter-defibrillator (ICD) is a device used to treat abnormal cardiac rhythms or placed as a prophylactic measure in patients who are at risk of such rhythms.
Implanted in the same fashion as a demand pacemaker, the ventricular lead has two defibrillator coils which can be seen on an X-ray (see below – arrows mark the coils) and can deliver internal defibrillation in response to VT or VF.
They can also perform anti-tachycardia pacing (ATP) in response to VT which is not immediately life-threatening to try and reduce the number of shocks. This involves pacing the ventricle for a short burst faster than the VT circuit, terminating the rhythm.
Most ICD’s select their therapy depending on pre-defined heart rate bands:
- Monitoring zone: the device will not take any action but will record the event
- VT zone: in this zone, the device will try ATP initially and then deliver a shock if a defined number of ATP cycles fail
- VF zone: If the rate is high enough, the device will assume the rhythm is fast VT or VF and is likely to be haemodynamically unstable. It will then begin delivering shocks as first-line therapy.
Whilst more modern devices can determine the underlying rhythm, patients with AF and a fast enough ventricular rate can receive inappropriate shocks as the device is triggered simply by the high ventricular rate.
These events can have a significant psychological impact. Patients describe an ICD discharge as like being ‘kicked by a horse’ and it is unlikely the internal cardioversion will terminate the rhythm resulting in multiple shocks.
Also, as fast AF is usually haemodynamically tolerated, patients are conscious throughout. Some patients can also develop VT or VF which does not terminate despite appropriate therapies or rapidly recurs. This can result in multiple shocks and is called a VT/VF storm.
Indications for insertion of an ICD
NICE guidance classifies patients who are candidates for ICD insertion with primary or secondary prevention indications.5
Secondary prevention
Those who have had a serious ventricular arrhythmia without a treatable cause:
- Survivors of cardiac arrest due to VT/VF
- Spontaneous sustained VT with syncope or haemodynamic compromise
- Sustained VT without syncope/haemodynamic compromise but with left ventricular ejection fraction (LVEF) < 35% and symptoms of NYHA III or better
An important feature of this category is the ‘without a treatable cause‘ caveat. Many patients with ST-elevation MI’s will present with VT or VF. However, this is clearly a treatable cause and opening the occluded artery will terminate the arrhythmia.
Similarly, VT due to electrolyte abnormalities will cease when they are corrected. These people do not need ICD’s unless the arrhythmia persists after the cause is corrected.
Primary prevention
Those who are at risk of serious ventricular arrhythmia:
- Familial cardiac conditions such as hypertrophic cardiomyopathy, Brugada syndrome, long QT syndrome or ARVC with high-risk features for sudden cardiac death
- Previous surgical repair of congenital heart disease
- Patients with previous myocardial infarction, LVEF < 35% and heart failure symptoms
Ventricular tachycardia definition
Ventricular tachycardia is defined as three or more beats originating from the ventricles in rapid succession.
Three to five beats is called a salvo. Non-sustained VT is defined as VT lasting more than 6 beats but less than 30 seconds.
Sustained VT is defined as VT that lasts more than 30 seconds or causes haemodynamic instability.
Cardiac resynchronisation therapy
In patients with LV dysfunction and a broad QRS complex (> 120ms), the walls of the ventricle can become uncoordinated or dyssynchronous.
Normally, the Purkinje fibres rapidly conduct the atrial impulses to both ventricles simultaneously and everything contracts in a coordinated synchronous fashion.
When a bundle branch is blocked, conduction heads down the functioning bundle to the corresponding ventricle, then slowly propagates across the myocardium to the other ventricle causing dyssynchronous contraction.
This dyssynchrony can significantly impact on the efficiency of the heart and, in the setting of LV dysfunction, can make heart failure symptoms significantly worse.
To correct this, cardiac resynchronization therapy was devised to pace the left and right ventricles together in response to atrial contraction to return the heart to as close to normal physiology as possible.
This is done by passing a third pacing lead into the coronary sinus and lodging it into a small cardiac vein (see the X-ray below for an example of a CRT-D device, the defibrillator coils can be seen on the ventricular leads).
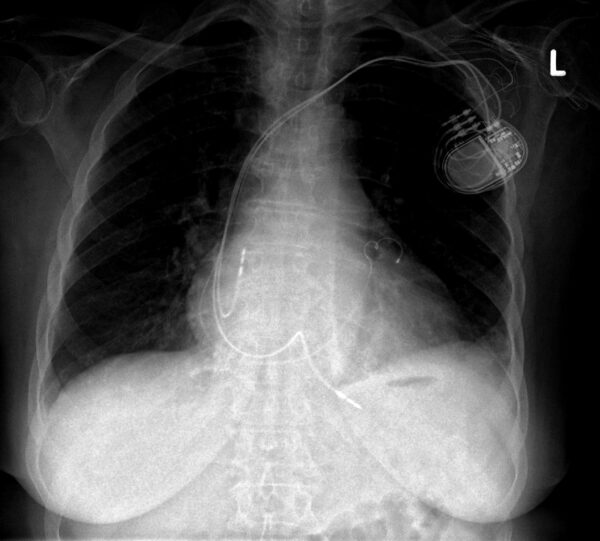
CRT devices work slightly differently to normal pacemakers. Rather than pacing only on demand, you want the patient to be using the pacemaker > 90% of the time to get the maximum effect from the device.
As such, the pacemaker’s base rate is set higher than demand pacemakers. The patient is then beta blocked to ensure their intrinsic rate is less than this, encouraging the pacemaker to work as much as possible. Similarly, if the patient is in AF, their rate will need to be well controlled to ensure they use the pacemaker as much as possible (if this is not possible, see the ‘pace and ablate’ entry below).
Special pacing scenarios
‘Pace and ablate’
This refers to the use of a pacemaker to treat atrial fibrillation with either an uncontrollable rate (whether due to intolerance of medication or simply because maximal doses of rate-limiting medication are not working) or uncontrollable palpitation symptoms.
A pacemaker is inserted, and the AV node is ablated leaving the patient dependent on the paced rhythm. This alleviates most palpitation symptoms patients feel but does mean that the patient will require a pacemaker for the rest of their life.
If the patient is 80, this means they may need one box change during their life (with the associated risk of infection) which is an acceptable risk for the benefits. However, if the patient is 30, they will need 6-8 box changes, meaning a significant amount of risk is involved with this management option.
Patients who have LV dysfunction and uncontrolled AF should receive a CRT device (to maintain physiological heart function) and then undergo AV node ablation leaving them pacing dependent, but with synchronous contraction.
‘Tachy-brady syndrome’
Pacemakers alone can only deal with bradyarrhythmias. In patients who have tachycardia and, while treating this rhythm, encounter problems with bradycardias (e.g. paroxysmal AF requiring large doses of beta-blockade to control the fast AF with secondary cardioversion to symptomatic bradycardia or heart block).
In this situation, the pacemaker is used as a safety measure to prevent bradycardias allowing you to use the large doses of rate-limiting medication required to control the tachycardia.
Pacemaker problems
Pacemaker generators are one of the most reliable medical devices in existence. However, there are several problems that can occur following the implantation of a permanent pacing system
Infection
Infected pacemakers must be removed. This includes infected pacemaker pockets or endocarditis affecting pacemaker leads. Any break in the skin over the pacemaker which reveals the metal underneath is also an indication for extraction.
Lead problems
Displacement
Unfortunately, leads can move and become displaced. An extreme version of lead displacement is called ‘Twiddler’s syndrome’ and typically occurs when patients idly play with their pacemaker causing the leads to be pulled out of the heart.
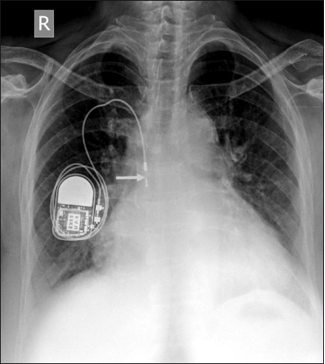
Fracture/subclavian crush
The leads pass below the clavicle and if there is limited space, leads can rub together or rub against the clavicle. This can cause damage to the leads.
Pacemaker mediated tachycardia
This rare phenomenon occurs when the AV node can conduct retrogradely (from V to A). In this situation, a re-entry circuit is set up with the pacemaker acting as the A to V connection and the native conduction carrying the impulse back up to the atria.
Fiddling with the pacemaker settings to increase the VA delay will sort this out so the pacemaker ignores the retrograde transmission.
Pacemaker syndrome
In some patients who are RV paced without an atrial lead, the AV node can conduct retrogradely. This causes dysynchrony between the atria and ventricles and can cause significant mitral and tricuspid regurgitation along with heart failure symptoms.
The solution is to place an atrial lead to re-synchronise the chambers or to adjust the pacemaker settings to minimise V pacing (difficult when the patient is pacing dependent!)
Additional information
Pacemakers and surgery
Pacemakers are safe in surgery. However, bipolar diathermy must be used to prevent the box from resetting to default.
ICD’s should be deactivated during surgery as diathermy is detected by the defibrillator as VF resulting in shock delivery.
Pacemakers and magnets
Placing a magnet over a pacemaker will set it to pace at 100bpm regardless of the underlying rhythm.
Placing a magnet over an ICD will turn off the shock function and is a good emergency measure if a patient is being repeatedly shocked or if the patient is dying. It does not disable the pacing function.
Pacemakers and driving
Patients with pacemakers cannot drive a car for 1 week or a group 2 vehicle (HGV/ bus) for 6 weeks after implantation. They then have no restrictions.
Patients with ICD’s cannot drive a car for 1 month after implant for primary prevention (i.e. without a preceding VT/VF event) or 6 months for secondary prevention (i.e. after a VT/VF event). They can then drive unless they receive a shock. At this point, they must take 6 months off driving. An inappropriate shock incurs a 1-month ban providing the cause of the inappropriate shock has been removed.
Patients with ICD’s cannot hold a group 2 licence (HGV’s/buses etc).
References
- NICE Guideline TA134. Implantable cardioverter-defibrillators and cardiac resynchronisation therapy for arrhythmias and heart failure. Published June 2014. Available from: [LINK]
- Pacemaker image. Steven Fruitsmaak. Licence: [CC BY 3.0]. Available from: [LINK]
- ECG showing pacing spikes. Michael Rosengarten BEng, MD.McGill. Licence: [CC BY-SA]. Available from: [LINK]
- Pacemaker appearance on a chest X-ray image. Stephanie C Torres-Ayala, Guido Santacana-Laffitte, and José Maldonado. Licence: CC BY 4.0. Available from: [LINK]
- ICD appearance on a chest X-ray image. PeaBrainC. Licence: [CC BY-SA]. Available from: [LINK]
- CRT device appearance on chest X-ray. Zimmermann. Licence: [CC BY-SA]. Available from: [LINK]




