- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
This guide provides a structured approach to HIV pre-exposure prophylaxis (PrEP) counselling in an OSCE setting. For more information on HIV, see the Geeky Medics guide.
Download the PrEP counselling PDF OSCE checklist, or use our interactive OSCE checklist.
Background
Indications for PrEP
PrEP is recommended in the following groups:
- Men who have sex with men (MSM) or trans women reporting condomless sex in the last 6 months and ongoing condomless anal sex.
- Anyone reporting condomless sex with someone who is known to be HIV positive, unless the partner has been on antiretroviral therapy (ART) for at least 6 months and their plasma viral load is <200 copies/mL.
PrEP should be considered for other patients on an individual basis.
There are many risk factors that should be taken into consideration including:
- Reporting future anticipated high-risk sexual behaviour
- Bacterial rectal sexually transmitted infection (STI) or hepatitis C (HCV) in the previous year
- Use of post-exposure prophylaxis (PEP) in the previous year
- Feeling unable to negotiate and/or use condoms
- Condomless sex with high-risk populations
- Injecting recreational drug use/sharing drug-taking equipment
Contraindications to PrEP
PrEP should not be taken by people who are HIV positive.
It is therefore important to ask about any recent episodes of condomless sex and subsequent seroconversion symptoms. PrEP is usually not required in those that report consistent condom use. PrEP is also often unnecessary if the patient only engages in sexual activity with HIV positive partners who are on treatment and have an undetectable viral load.
Opening the consultation
Wash your hands and don PPE if appropriate.
Introduce yourself to the patient including your name and role.
Clarify the purpose of the consultation: “I believe that you would like to discuss pre-exposure prophylaxis today/PrEP today, is that correct?”
It is important to establish rapport and an open line of communication: “If at any point you have questions or feel that I have not clearly explained something please feel free to interrupt me so that I can clarify.”
Patient’s ideas concerns and expectations
An important part of counselling is eliciting and exploring a patient’s ideas, concerns, and expectations.
Asking about a patient’s ideas, concerns, and expectations can allow you to gain insight into how a patient currently perceives their situation, what they are worried about and what they expect from the consultation. It can sometimes be challenging to use the ICE structure in a way that sounds natural in your consultation, but we have provided some examples for each of the three areas below.
Ideas
- “What do you know about PrEP so far?”
- “Have you read or heard about PrEP before?”
- “What are your thoughts about PrEP?”
Concerns
- “Is there anything that worries you about taking PrEP?”
Expectations
- “What were you hoping to get from our discussion today?”
- “Is there anything, in particular, you would like me to cover today?”
What is PrEP?
PrEP or pre-exposure prophylaxis is a combination of two antiretroviral drugs, Tenofovir DF and Emtricitabine. These drugs have been widely used to treat HIV for many years. They work by preventing HIV from multiplying in T cells.
There have been multiple trials assessing the effectiveness of PreP. These trials demonstrated a significant reduction in HIV infection in those prescribed PrEP versus those not.
PrEP reduces the chance of being infected with HIV via sexual intercourse by up to 99% if it is taken as prescribed.
Example
“PrEP stands for pre-exposure prophylaxis. It is a medication that you take before and after sex which can help prevent you from being infected with HIV.”
“It is a combination of two medications which stop the virus infecting your cells. We have been using these drugs to treat HIV for a long time.”
“Studies have shown that if PrEP is taken reliably, it reduces the chance of being infected with HIV via sexual intercourse by up to 99%.”
How to take PrEP
There are two ways in which PrEP can be taken. The first is by taking a single pill every day which is known as daily dosing. The second is by taking tablets prior to and after sexual contact, which is known as event-based dosing.
Daily dosing
Most studies of PrEP were conducted using the daily dosing method. Daily dosing involves taking PrEP every day.
Taking PrEP every single day results in a consistent level of protection from HIV infection at all times provided it is taken correctly. This means that sexual encounters are not something that would need to be planned for.
Daily dosing is appropriate for anyone who is starting PrEP.
The time to protection varies depending on the type of sex:
- Anal sex: initial protection is provided by taking two pills 2 to 24 hours before sex and then one subsequent pill at 24 and 48 hours after (as with event-based dosing described below). A daily pill from 72 hours onwards then provides future protection, as long as there is no break.
- Vaginal sex: the time to clinical protection is 7 days of daily dosing.
Event-based dosing
Event-based dosing involves taking PrEP before sex and for a period after sex.
It is appropriate for anal sex and men having insertive vaginal sex. However, if a patient is taking more than four tablets a week on a regular basis, daily dosing may be a simpler option.
Event-based dosing is contraindicated in anyone having receptive vaginal sex or with a current hepatitis B infection.
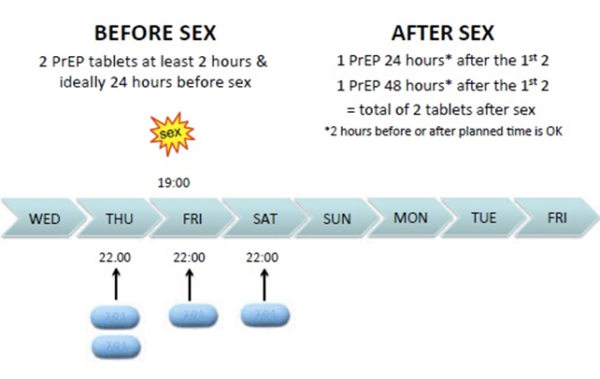
For event-based PrEP to be effective it should be taken in the following way:
- Two tablets between 2 and 24 hours before sex
- A single pill 24 hours after the first dose
- Another pill 24 hours after the second dose
Therefore, a double dose should be taken prior to sex. Then a single dose again at both 24 and 48 hours (Figure 1).
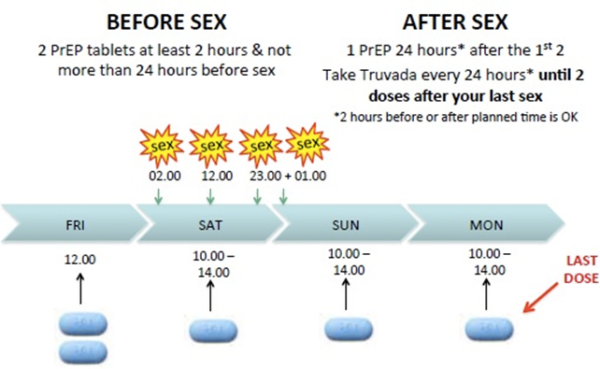
If a patient is planning to have sex multiple times over a period of more than 24 hours, they should continue to take PrEP every 24 hours until 48 hours have passed since the last episode of sex (Figure 2).
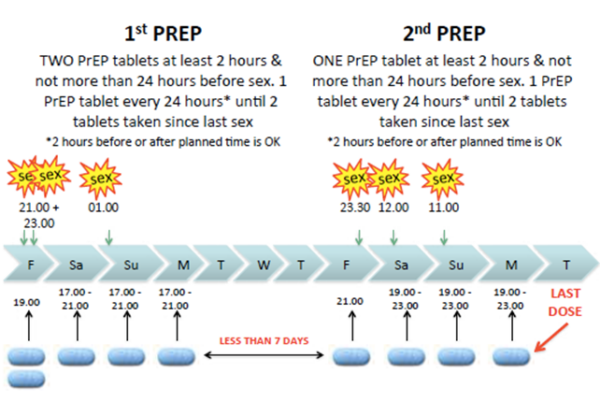
If a patient plans to have sex less than a week after they last finished PrEP, they can take a single initial dose rather than a double dose (Figure 3).
Side effects
Common side effects of PrEP include nausea, headache, and gastrointestinal disturbance. These are normally mild and usually settle within the first two weeks.
Occasionally PrEP can cause more serious side effects. In some patients, it can result in a reduction in creatinine clearance. When this does occur, it tends to be mild, non-progressive, and reversible.
Small decreases in bone density have also been noted in some patients taking PrEP, but there is no evidence of increased fracture risk.
Example
“As with all medications, there is a risk of potential side effects. Common ones include headache, nausea, or an upset stomach. These tend to settle in a few weeks but if you have any difficulties please contact us.”
“There is a chance that PrEP can affect your kidneys and bone density, so we’ll need to monitor your kidney function with regular urine and blood tests. If there are any issues, these typically resolve after stopping the medication.”
Monitoring
Before starting PrEP several baseline tests are required along with regular monitoring every three months.
HIV testing
PrEP can only be taken if an individual is HIV negative.
The drugs used in PrEP are also used to treat HIV. However, when treating HIV, a combination of drugs is used. This means PrEP would only partially treat a HIV infection, putting the individual at risk of developing resistance to medications.
Fourth-generation HIV tests have a window period of 45 days. If a patient has had condomless sexual intercourse within the last 45 days, they can start PrEP if they are at high risk of contracting HIV providing they have no symptoms of seroconversion illness and are aware of the potential risk.
However, patients should be asked to return as soon as they are outside of the window period for a repeat HIV test.
For all patients, a repeat HIV test should be performed every 3 months throughout treatment.
Hepatitis B
Tenofovir, one antiviral within PrEP can also be used to treat hepatitis B. Should a patient test positive for HBV they can take daily PrEP under the supervision of a hepatology service. However, event-based dosing would not be an option. This is because withdrawal of PrEP in HBV positive individuals can cause rebound viraemia and fulminant liver damage. All HBV negative individuals should be offered vaccinations against hepatitis B.
Renal function
Given that PrEP can affect renal function, baseline urea and electrolytes (U&Es) should be taken and repeated at least annually. A urine dipstick to check for protein should be performed every three months and if positive followed up with additional U&Es.
Routine STI testing
Routine STI testing is recommended every three months in all patients, but particularly those who have had multiple sexual partners in the previous window.
Routine STI screening involves testing for:
- Chlamydia
- Gonorrhoea
- Syphilis
- HIV
- Hepatitis C: for individuals identified as at risk
- Hepatitis A: for at-risk individuals, a vaccine is also available
For more information, see the Geeky Medics guide to sexually transmitted infections.
Example
“Before you start PrEP it is important we do some baseline blood tests to ensure that it is safe for you to take it.”
“If you start PrEP and already have HIV it can lead to the virus becoming resistant to the medication. This means that some of the drugs we might use to treat would not work as well.”
“We need to do a blood test to look for a virus called hepatitis B. If you were found to be infected with hepatitis B, event-based dosing wouldn’t be suitable for you and we would need to seek advice from the liver team about treatment. If you are found to not be infected with the hepatitis B virus, we can offer you a vaccine against it to protect you in the future.”
“We know that occasionally PrEP can affect the way your kidneys work. It is important that we know how well they are working today so we can quickly identify any deterioration in the future.”
“We recommend that everybody who is sexually active gets a regular sexual health screen every 3 months, especially if they are having condomless sex.”
Missed pills
PrEP is most effective when levels in the body are above a certain threshold level. It is therefore important to encourage patients to adhere to treatments. However, missed doses can easily happen and so patients should be advised to seek advice should this occur.
Daily dosing
If one or two doses are missed, then this is not an issue providing it has been taken consistently for at least seven days.
However, repeatedly missed doses result in a decreased level of protection. Studies show that if a patient misses three doses in a one-week period, protection drops to 96%. This falls further to 76% if five pills are missed.
If a patient has missed more than seven pills in a row they should restart with a double dose and then continue with a single dose from the next day onwards.
Event-based dosing
If the first double dose is forgotten before sex it should be taken immediately after and then a single pill continued daily. The patient should also be advised to contact their local clinic or emergency department to discuss the need for post-exposure prophylaxis (PEP).
Example
“If you miss a pill occasionally this should not cause an issue providing you have taken it consistently for at least one week prior. However, if you are missing multiple pills in a week then you will not be as well protected. If this is the case please contact us as you may be eligible for post-exposure prophylaxis which is also known as PEP.”
“If you forget to take your double dose before sexual contact, take it as soon as you remember and carry on with a single dose each day afterwards. Any PrEP is better than no PrEP. However, if this does happen, we recommend contacting a doctor as you may need PEP.”
PrEP in specific groups
Whilst PrEP is most commonly used amongst men who have sex with men, it is also suitable for numerous other groups who may have slightly different questions or needs.
Specific advice for PrEP in women includes:
- PrEP will not affect contraception and vice versa.
- Studies have not shown any pregnancy complications from PrEP. However, the sample sizes were small so women should be encouraged to speak with their doctor.
- Daily PrEP can be used safely when breastfeeding.
Specific advice for PrEP in trans and non-binary people:
- If having vaginal sex daily dosing is needed.
- If only having anal sex then event-based dosing can also be used.
- PrEP will not affect any hormone treatment being taken.
Closing the consultation
Close the consultation by summarising what you have discussed. This allows you to emphasise the key points of the consultation.
Finally, thank the patient for their time and offer them a leaflet summarising the key information related to PrEP.
Dispose of PPE appropriately and wash your hands.
Example
“Before we finish I would like to check that we have been through everything you need. We have discussed how PrEP works, how to take it, what to do if you miss a dose, common side effects, and follow-up going forwards. Is there anything you feel we have missed? Or anything else you would like to ask?”
Reviewer
Dr Andrew Tomkins
Consultant in Genitourinary Medicine
North Manchester General Hospital
Editor
Dr Chris Jefferies
References
- Anderson, P.L., Glidden, D.V., Liu, A., Buchbinder, S., Lama, J.R., Guanira, J.V., McMahan, V., Bushman, L.R., Casapia, M., Montoya-Herrera, O., Veloso, V.G., Mayer, K.H., Chariyalertsak, S., Schechter, M., Bekker, L.-G. ., Kallas, E.G. and Grant, R.M. (2012). Emtricitabine-Tenofovir Concentrations and Pre-Exposure Prophylaxis Efficacy in Men Who Have Sex with Men. Science Translational Medicine, [online] 4(151), pp.151ra125–151ra125. Available from: [LINK]
- Brady, M., Rodger, A., Asboe, D., Cambiano, V., Clutterbuck, D., Desai, M., Field, N., Harbottle, J., Jamal, Z., McCormack, S., Palfreeman, A., Portman, M., Quinn, K., Tenant-Flowers, M., Wilkins, E. and Young, M. (2018). BHIVA/BASHH guidelines on the use of HIV pre-exposure prophylaxis (PrEP) 2018. [online] Available from: [LINK]
- Figure 1. Figure 2. Figure 3. i-base.info. (2019). UK guide to PrEP | Guides | HIV i-Base. [online] Available from: [LINK]
- Molina, J.-M., Capitant, C., Spire, B., Pialoux, G., Cotte, L., Charreau, I., Tremblay, C., Le Gall, J.-M., Cua, E., Pasquet, A., Raffi, F., Pintado, C., Chidiac, C., Chas, J., Charbonneau, P., Delaugerre, C., Suzan-Monti, M., Loze, B., Fonsart, J. and Peytavin, G. (2015). On-Demand Preexposure Prophylaxis in Men at High Risk for HIV-1 Infection. New England Journal of Medicine, 373(23), pp.2237–2246.
- PrEP Impact Trial. (2017). PrEP | The PrEP Impact Trial. [online] Available from: [LINK]
- Rodger, A.J., Cambiano, V., Bruun, T., Vernazza, P., Collins, S., Degen, O., Corbelli, G.M., Estrada, V., Geretti, A.M., Beloukas, A., Raben, D., Coll, P., Antinori, A., Nwokolo, N., Rieger, A., Prins, J.M., Blaxhult, A., Weber, R., Van Eeden, A. and Brockmeyer, N.H. (2019). Risk of HIV transmission through condomless sex in serodifferent gay couples with the HIV-positive partner taking suppressive antiretroviral therapy (PARTNER): final results of a multicentre, prospective, observational study. The Lancet. Available from: [LINK]




