- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
The coagulation screen is an important diagnostic test, yet is notorious for misinterpretation. In order to make the coagulation screen easier to interpret, it is important to understand the physiology of what is being assessed.
Why do we perform this test?
The main reasons for performing a coagulation screen are:
- To confirm a suspected coagulopathy.
- To monitor the coagulation status of a patient on long/short term anti-coagulants.
- To assess a patients coagulation status prior to surgery.
- To assess the synthetic function of the liver.
How does coagulation work?
The coagulation screen assesses the function of the clotting cascade and the body’s ability to create a platelet plug. First, we shall look at how platelets work.
Platelets
In response to vascular injury, the following steps lead to a platelet plug:
1. von Willebrand’s Factor (vWF) adheres to the vascular injury.
2. vWF then binds to GpIb receptors on surrounding platelets.
3. After binding, the ADP (P2Y12) receptor on the platelet is activated leads to increased expression of GpIIb/IIIa.
4. These newly expressed receptors then finally bind to fibrinogen, which leads to further platelet aggregation.
Now, let’s look at how the clotting cascade works.
Clotting cascade
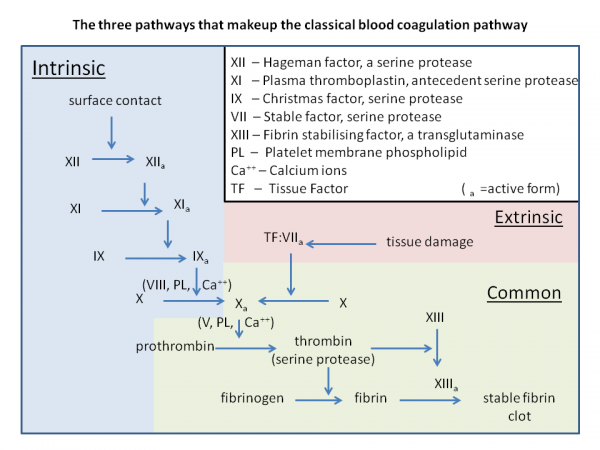
The clotting cascade is comprised of many different factors, ranging from I through to XIII. These factors form 3 groups: the intrinsic pathway, the extrinsic pathway, and the combined pathway.
Key features of each pathway are shown below.
Intrinsic pathway
The intrinsic pathway is activated by contact with a damaged surface and goes as follows:
Factor XII → Factor XI → Factor IX (+ Factor VIII + vWF) → Common pathway
Extrinsic pathway
The extrinsic pathway is activated by the release of tissue factors and goes as follows:
Factor VII → Common pathway
Combined pathway
The combined pathway then proceeds the intrinsic/extrinsic pathways and goes as follows:
Factor X (+ Factor V) → Factor II → Factor I (+ Factor XIII → Fibrin mesh)
The diagram below should help put all of this into context.
How are clots later dissolved?
Fibrin clots can then be dissolved through a separate pathway whereby plasminogen becomes its activated form, plasmin, via tPA. This plasmin then breaks down the fibrin mesh.
Tests
There several components to the coagulation screen, some of the core parts and what they mean are covered below.
PT/INR (12-13 seconds/0.8-1.2)
The prothrombin time (PT) is a measure of the time taken for blood to clot via the extrinsic pathway (a good way to remember is that you ‘Play Tennis OUTSIDE’ therefore PT is EXTRINSIC).
International normalised ratio (INR) is a standardised version of this test, commonly used with patients on anticoagulants.
Since the only factor in the pathway is VII (which is rarely deficient in isolation), these tests are a measure of overall clotting factor synthesis or consumption.
This test can be affected by liver disease, disseminated intravascular coagulation (DIC), vitamin K deficiency and warfarin levels.
APTT (35-45 seconds)
The activated partial thromboplastin time (APTT) is a measure of the time taken for blood to clot via the intrinsic pathway (a good way to remember, following PT, is that you ‘Play Table Tennis INSIDE’ therefore PTT is INTRINSIC).
Like with PT, APTT time will be affected by overall clotting factor synthesis or consumption (it can also be affected by DIC, liver failure, vitamin K deficiency and warfarin levels).
APTT, however, can indicate issues with factors VIII (and vWF), IX, and XI specifically.
The main conditions that could result in an abnormal APTT include:
- Haemophilia A (VIII – X-linked recessive)
- Haemophilia B (IX – X-linked recessive)
- Haemophilia C (XI – autosomal recessive)
- von Willebrands disease (as vWF pairs up with factor VIII)
Note: anti-phospholipid syndrome can cause a high APTT despite being a disorder that causes clots, due to it inactivating the phospholipid used in APTT.
Bleeding time (1-6 minutes for finger prick)
Whilst no longer formally used due to its rather barbaric nature, bleeding time assesses overall platelet function and levels. It is therefore worth noting the time it takes for a patient to stop bleeding from any cuts.
As such, platelet specific disorders will increase the overall bleeding time.
Some examples of such disorders that will increase bleeding time include:
- von Willebrand’s disease (vWF deficiency – autosomal dominant)
- Bernard-Soulier syndrome (GpIb deficiency)
- Glanzmann thrombasthenia (GpIIb/IIIa deficiency)
- TTP/ITP/HUS/DIC
- Thrombocytopaenia
Thrombin time (10-15 seconds)
This is a test of how fast fibrinogen is converted to fibrin by thrombin.
In cases of a prolonged time, the cause is either a synthetic issue or consumption issue.
Similar to prolonged PT, this can be due to DIC, liver failure, malnutrition, abnormal fibrinolysis and many other conditions.
Extra tests
Further tests can be ordered to work out the exact pathology. These can provide levels of specific factors, antibodies, thrombophilia screens, ADAMTS13 (for TTP) and many more – however, some of these can only be ordered at the request of a haematologist.
It is also worth ordering a full blood count (to assess platelet levels), liver function tests (to assess for general liver function abnormalities), albumin (to assess the liver’s synthetic function/malnutrition) and D-dimer.
What are the patterns of some common coagulopathies?
| Condition | PT/INR | APTT | BT | Platelet count | Notes |
| Vitamin K deficiency/ Warfarin use | ↑ | ↑ | – | – | Different INR required in the management of different conditions, so a high value may be normal for the patient (e.g. AF, valve replacements, etc) |
| Haemophilia A/B/C | – | ↑ | – | – | Symptoms of ‘clotting’ disorders: haemarthrosis, muscle haematomas, prolonged bleeding after dental surgery |
| von Willebrand’s disease | – | -/↑ | ↑ | – | Symptoms of ‘platelet’ disorders: petechiae, bruising, contact bleeding (e.g. gums), menorrhagia |
| DIC | ↑ | ↑ | ↑ | ↓ |
Total coagulopathy, primary cause must be treated, give platelets and clotting factors
|
| ITP/TTP/HUS | – | – | ↑ | ↓ |
Lack of PT/APTT derangement is a good differentiator from DIC, NEVER give platelets to these patients
|
What are the effects of anticoagulants and how do I take this into account?
All anticoagulants (warfarin, heparins, NOACs) will increase the PT/INR and the APTT; this should, therefore, be considered when interpreting results. Patients on warfarin will have a specific target INR depending on the condition they have (AF, valve replacements, etc), it is, therefore, a good idea to get a medical history from the patient.
Antiplatelets, such as clopidogrel (P2Y12 inhibitor) and aspirin, will also cause derangement. These, however, will cause an increase in the overall bleeding time but won’t affect PT or APTT.
What key points should I take away from this?
PT/INR assesses the EXTRINSIC pathway – since factor VII pathology is rare, it is better used as a measure of overall clotting. It will, therefore, be affected in anti-coagulant use, liver failure, and DIC
APTT assesses the INTRINSIC pathway – this, therefore, measures factors VIII (and vWF), IX, and XI. The most common causes of increased APTT are haemophilia A (VIII), B (IX), C (XI), and possibly von Willebrand’s disease (since vWF pairs with VIII)
Bleeding time – although no longer used formally – measures the formation of the platelet plug. This will be affected by platelet disorders like von Willebrand’s disease (vWF), Bernard-Soulier syndrome (GpIb), TTP, ITP, HUS, and thrombocytopaenia.
Total clotting derangement is rare, but the cause must be identified quickly (e.g. DIC, malnutrition, or liver failure).
Make sure to order complementary tests: FBC and LFTs can provide greater insight into a possible cause when correlated with the results of the coagulation screen.
References
Texts
- Hall, J. & Guyton, A. Guyton and Hall textbook of medical physiology (13th Edition). (Saunders Elsevier, 2011).
- Wilkinson, I., Raine, T. & Wiles, K. Oxford handbook of clinical medicine (9th Edition). (Oxford University Press, 2010).
- Le, T., Bhushan, V., Sochat, M., Petersen, M. & Micevic, G. First Aid for the USMLE Step 1 2019. (McGraw-Hill Education, 2019).
- Rang, H. P., Dale, M., Flower, R. J. & Henderson, G. Rang and Dale’s pharmacology (8th Edition). (Elsevier/Churchill Livingstone, 2016).
Images
- Dr Graham Beards. Coagulation pathway. Licence: [CC BY-SA 3.0]. Available from: [LINK].




