- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
Medication overdose is an increasingly common presentation, particularly in the emergency department, with patients presenting with a broad range of clinical features.
Of all drugs taken in overdose, opioids have been consistently shown to be the most likely to cause death.1
In 2018 4,359 deaths in England and Wales were attributed to medication overdose, 2,208 of which were due to opioid drugs (excluding paracetamol containing compounds, such as co-codamol).1
Opioid abuse is a global problem. The World Health Organisation (WHO) estimates that 500,000 deaths worldwide are attributed to drug use, with more than 21% of these attributed to opioid overdose. In 2017, the WHO estimates that 115,000 people died from opioid overdose.2
Aetiology
Terminology
The terms opiate and opioid are often used interchangeably. However, they have different meanings:
- An opiate is a naturally occurring alkaloid drug derived from the opium poppy (e.g. morphine and codeine).
- An opioid is any synthetic or semi-synthetic drug derived from the opium poppy (e.g. fentanyl and oxycodone).
Opioid is also used as the broad term for any substance which binds opioid receptors to produce an opiate-like toxidrome. All opiates are opioids, but not all opioids are opiates.3
Pharmacology
Opioid receptors are G-coupled proteins. The highest concentration of opioid receptors is in the central nervous system however some receptors exist in the peripheral nervous system.
There are three main receptor subtypes:
- Mu receptors
- Kappa receptors
- Delta receptors
Activation of all three receptor sub-types produces analgesic effects. This the main therapeutic reason for prescribing an opioid in a medical setting.
However, the side effects of receptor activation include respiratory depression, reduced consciousness, miosis (constricted pupils) and higher cortical processing effects such as euphoria and dependence.
The physiological roles (although not yet fully understood) of each receptor subtype are summarised below:4
- Mu receptors: this is the most abundant and best-understood receptor sub-type. These receptors have the highest affinity for morphine and produce the most potent analgesic effects by activating the mesolimbic dopaminergic system which is also responsible for euphoria, respiratory depression and constipation associated with opioid use.
- Kappa receptors: these receptors produce less analgesia than mu receptors. They are associated with reduced consciousness, respiratory depression, dependence and dysphoria.
- Delta receptors: the effects of Delta receptors have been least studied and are the least well understood; they are thought to be involved in the psychological effects of opioids including dysphoria.
Opioid drug examples
The ‘strength’ of an opioid drug is governed by its ability to act as an agonist at the mu receptor (as these produce the greatest analgesic effect) and its potency.
Table 1. The main medicinal forms of opioids.
| Generic name Strength |
Routes | Indications | Cautions |
| Codeine Weak |
Oral | Useful for most types of pain and in most patients. Also, in a form with paracetamol 8/500 (8mg codeine, 500mg paracetamol) available over the counter, 30/500 by prescription only. |
Generally quite constipating. |
| Tramadol Weak |
Oral IV |
Useful in postoperative analgesia for general surgery; less constipating than other opioid preparations and available parenterally. |
|
| Morphine Strong |
Oral, IV, SC | Easy to use for immediate severe pain relief and is familiar to most medical and nursing staff. IV dose can be titrated to effect in the immediate setting. Easy to titrate oral dose to requirement. |
|
| Fentanyl Strong |
IV, transdermal | Chronic pain treatment in patch form, an anaesthetic agent in IV form. | Extremely potent – prescribed in micrograms unlike most other opioids (in milligrams). |
| Methadone Strong |
Oral | A long-acting drug used in opioid addiction for weaning and avoiding withdrawal. |
|
| Heroin Strong |
IV | An illegal street drug, crude preparation of diamorphine. | The most common cause of death in opioid overdose.1 |
This table is not an exhaustive list of available opiates. Some preparations/routes are for specialist use only, for example fentanyl in any form.
Risk factors
There are different sub-groups of overdose and the risk factors are different for each.
Recreational drug use:
- Most commonly by intravenous drug users
- Any route of opioid administration has the potential to be abused
Intentional overdose:
- Self-harm or suicide attempt
- Less commonly an attempt to harm someone else in a person’s care, by purposefully administrating more than the prescribed dose
Unintentional overdose:
- Chronic pain or palliative care patients
- Elderly, more likely if starting a new opioid, changing a dose or starting a new interacting medication
- Children (safeguarding issue)
- Starting a new opioid, especially in the opioid naïve
- A therapeutic error by incorrect prescription or incorrect dose/form/agent administration
- Hepatic or renal impairment: opioid may not be metabolised fully or can accumulate to toxic levels
- CYP2D6 gene duplication carriers causing ultra-fast metabolism of codeine into its active metabolite, morphine, into potentially toxic concentrations5
Clinical features
History
In clinical practice, the presentation of opioid overdose is a spectrum ranging from mild symptoms to a completely comatose patient.
Many patients will present with a decreased level of consciousness and no specific symptoms. However, if symptoms are present these may include:
- Nausea and/or vomiting
- Constipation
- Pruritis
- Tiredness and increased somnolence
- Confusion (especially in the elderly)
Be aware these symptoms can also represent side effects in therapeutic non-overdose of opioid use, especially in the opioid-sensitive and/or opioid naïve patient subgroup.
Other important areas to cover in the history include:
- Drug history: obtain an accurate history of what opioids the patient is taking and compare this to their prescription. Be sure to review all sources of opioid prescription. It is an easy mistake for prescribers in different areas and systems (e.g. a GP and a hospital doctor) to prescribe opioids without realising the other already has.
- Past medical history: assess for risk factors for unintentional overdose (e.g. renal impairment)
- Psychiatric history: previous episodes of overdose and/or self-harm.
Mixed drug overdoses
Patients often present with mixed drug overdoses. Be sure to take a thorough history and formulate a management plan for all ingested substances. Consult local poisons information services (e.g. Toxbase) if required.
Clarify the preparation of opioid your patient has taken; some opioids come in paracetamol containing preparations, for example, co-codamol and co-dydramol. It is imperative to investigate and treat for paracetamol overdose, due to the risk of long-lasting and potentially fatal hepatic complications.
Unfortunately, the patient may have overdosed as an attempt to self-harm or end their life. Be sure to complete a thorough risk assessment and mental state exam as part of your assessment.
Clinical examination
The classical clinical toxidrome of opioid overdose is a triad of:
- reduced consciousness
- respiratory depression
- miosis
As with all acutely unwell patients, clinical assessment should follow an ABCDE approach.
Airway
The patient may be awake and talking with or without slurred speech, demonstrating that their airway is patent. In more severe overdoses, because of decreased consciousness, the airway may become partially obstructed with snoring or noisy breathing.
Partial airway obstruction can rapidly lead to complete airway obstruction and cardiorespiratory arrest. Be aware that vomiting in a patient with a decreased level of consciousness conscious poses a potential risk to the airway.
Breathing
Respiratory depression in opioid overdose presents with bradypnoea, hypoxia and in severe cases respiratory arrest.
A low respiratory rate (<12 breaths/minute) is an adverse sign and requires immediate medical attention.
Circulation
Patients typically present with bradycardia. Opioids, especially in IV form, cause vasodilation and hypotension.
Disability
Miosis, more commonly referred to as ‘pinpoint pupils’ and a decreased Glasgow Coma Scale (GCS) are clinical features of opioid overdose.
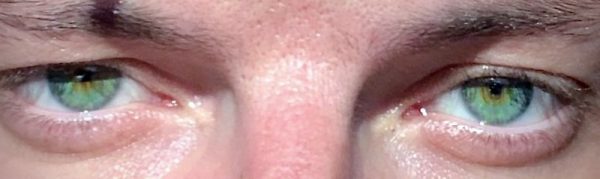
“Below 8, intubate”: patients with a GCS score less than 8, of any cause, are unable to protect their own airway, therefore, are at risk of obstructing it. They may require emergency intubation if the problem cannot be corrected quickly.
Exposure
Look for evidence of recreational drug use, for example, track marks on the arms. If the patient is in chronic pain or an end of life patient, they may have a syringe driver or a fentanyl patch which may easily be missed.
Look for evidence that would support other causes of reduced consciousness/respiratory depression, for example, intracranial pathology.
Take extra care in examining these patients. There may be drug paraphernalia around them/in pockets/hidden in clothing such as dirty needles.
Differential diagnoses
Reduced level of consciousness is a broad and challenging presentation with multiple differential diagnoses.
Important differential diagnoses to consider include:
- Hypoglycaemia: should be actively excluded in any patient presenting with reduced consciousness. Hypoglycaemia can be quickly diagnosed with a blood glucose reading and easily treated. The patient may have adrenergic (sweating, clamminess) and neuroglycopenic symptoms (confusion, drowsiness, aggression).
- Post-ictal status: a temporary state of confusion and depressed consciousness occurring after an epileptic seizure, typically lasting up to an hour, or longer in severe seizures.
- Overdose with other depressants: most commonly alcohol. Others include hypnotic drugs such as zopiclone, or benzodiazepines such as diazepam.
- Head injury or other intracranial pathology: often obvious from the collateral history or paramedics’ handover. Look for evidence of head injury for example lacerations and bruising, or from the history, for example, sudden onset occipital headache then loss of consciousness (typical of subarachnoid haemorrhage).
- Carbon dioxide narcosis: a state of reduced consciousness caused by carbon dioxide poisoning, typically occurs in those with chronic type 2 respiratory failure, for example, patients with chronic obstructive pulmonary disease. The patient will often feel warm and vasodilated with a bounding peripheral pulse.
Investigations
Bedside investigations
Relevant bedside investigations include:
- Observations (vital signs): to see if this patient needs urgent intervention and physiological support, for example, supplemental oxygen for low saturations.
- Arterial blood gas: to assess for evidence of type 2 respiratory failure and acidosis which may occur in opioid overdose secondary to respiratory depression.
- Capillary blood glucose: to rule out hypoglycaemia.
Bloods
Relevant blood tests include:
- FBC: important to obtain a baseline.
- U&E: to check if this patient is unknowingly accumulating opioids in their system due to renal impairment.
- LFTs: to assess if the patient can metabolise their opioid load effectively. Also useful to screen for acute liver damage secondary to overdose (e.g. mixed overdose with paracetamol). Chronically elevated liver enzymes may indicate viral hepatitis (often associated with intravenous drug use).
- Paracetamol levels: to rule out mixed overdose.
Paracetamol levels
It is considered good practice to take baseline blood tests (FBC, U+E, LFTs) and paracetamol levels from any patient presenting with overdose, regardless of the ingested substance.
A patient who intends to end their life, has drunk alcohol with their overdose (very common), and/or is drowsy or confused may not inform you of a potentially fatal paracetamol overdose, which has very few acute clinical features.
Imaging
A CT head should be performed if the patient’s level of consciousness does not improve after initial management (e.g. no response to Naloxone) to rule out intracranial pathology (should be discussed with a senior clinician first).
Management
Acute management
Removal of source
If possible, without endangering yourself or others, remove the source of opioid overdose (e.g. transdermal patch or syringe driver).
Monitoring
Patients who have overdosed on opioids have the potential to deteriorate rapidly (e.g. due to airway obstruction, respiratory arrest etc) and therefore should be monitored closely in an appropriate setting (e.g. monitoring bay).
Medical
You should assess and manage and life-threatening emergencies as you encounter them as part of an ABCDE approach. See the Geeky Medics article here for more detailed information.
Many patients require airway management and even ventilation support; this may be temporary until their opioid overdose is reversed, or for a longer time in a critical care unit if this cannot be achieved in a timely manner.
The mainstay of treatment of opioid overdose involves the use of a competitive opioid receptor antagonist such as naloxone.
Naloxone can be administered via intravenous, intramuscular, subcutaneous, intranasal, and oral routes. The oral route is used in the treatment of opioid-induced constipation and not in the acute setting.
Naloxone can be used as a diagnostic tool in the unconscious patient where the cause of loss of consciousness is unclear. A response to naloxone, represented by an improvement in the GCS score, miosis and respiratory rate, indicates the presence of opioid overdose. Alternative diagnoses should be considered if there is no response to naloxone.
Opioid reversal with naloxone is almost instant if given in sufficient doses, however, the effects can be short-lived. The half-life of naloxone is 60-90 minutes whereas the half-life of most opioids is significantly longer, which can result in initial improvement followed by recurrence of opiate overdose features if further doses of naloxone are not administered.6 As a result, patients need to be closely monitored for an extended period until the opiates in their system have been cleared.
Naloxone is commonly administered in repeat intravenous (IV) boluses titrated to effect. The IV route provides the most rapid onset of action and the ability to easily titrate doses.
However, there are times when different routes and methods of naloxone administration may be indicated:
- Intravenous drug users: there is a risk that once overdose has been reversed (or partially reversed), these patients will abscond only to relapse into a coma as the antagonist effect of the naloxone wears off and the effects of the opioids re-emerge. A simultaneous intramuscular (IM) dose of naloxone may be administered with the IV dose as a means of reducing the likelihood of this occurring.
- Chronic pain or palliative care patients: it is not desirable to fully reverse the effects of the opioids in this group of patients as you risk causing significant distress and pain. Naloxone should be administered in small aliquots, titrating the dose to reverse the toxidrome without reversing all the desirable analgesic effects.
- High opioid load: sometimes patients have such a high opioid load (e.g. a large overdose, reduced renal excretion) that it is difficult to manage with intermittent boluses of naloxone. These patients may be commenced on a naloxone infusion until they have cleared adequate amounts of opiate from their system.
- At home/out of hospital use: In the United Kingdom, Public Health England has updated their regulations, which now allow any person from a ‘drug treatment service’ to provide naloxone (e.g. recreational drug users at risk, hostel managers and police officers).7 Chronic pain and palliative care patients often have home naloxone kits for self or assisted administration in case of accidental overdose.
Long term management
Patients presenting with an intentional overdose will require mental health assessment and the involvement of psychiatric services if appropriate. If the patient has children at home, social services may need to be involved.
Patients presenting with unintentional overdose require a thorough review of their current medication regime and education on the risks of opiate overdose. If the patient was using recreational drugs, they will require referral to local drug cessation services.
Complications
Opioid overdose
Untreated significant opioid overdose will ultimately result in death secondary to respiratory depression. This is the major cause of mortality in opioid overdose.
Acute lung injury following heroin overdose is a well-recognised complication and presents similarly to acute respiratory distress syndrome (ARDS).
Naloxone treatment
Naloxone is an uncomplicated drug with a very safe side effect profile. The most commonly reported complications are the precipitation of acute withdrawal syndrome, and nausea and vomiting although these are usually short-lived.7
Key points
- Opioids are the commonest cause of overdose-related deaths (in England and Wales).
- Opioids bind Mu, Kappa and Delta receptors in the central and peripheral nervous system providing an analgesic effect.
- At-risk groups include recreational drug users, chronic pain and palliative care patients, those with self-harm and/or suicidal intent, and victims of therapeutic error.
- The classical clinical features of opioid overdose include the triad of respiratory depression, reduced consciousness and miosis.
- The key investigation in opiate overdose is an arterial blood gas to assess the degree of type 2 respiratory failure.
- Acute management involves the use of naloxone an opioid-receptor antagoninst.
- Assessment of the response to naloxone is a useful tool for the diagnosis of opioid overdose in the unconscious patient.
Reviewer
Emergency Medicine Consultant
Editor
Dr Chris Jefferies
References
- Office for National Statistics. Deaths related to drug poisoning by selected substances. Published in 2019. Available from: [LINK].
- World Health Organisation. Opioid overdose. Published in 2020. Available from: [LINK]
- A Rosenblum et al. Opioids and the treatment of chronic pain: Controversies, current status, and future directions. Published in 2008. Available from: Experimental Clinical Psychopharmacology, 16(5), 405-416.
- V Sharma et al. Opioid pharmacology: a review. Published in 2015. Available from: International journal of scientific research in science and technology, volume 1, issue 5.
- J Kirchheiner et al. Pharmacokinetics of codeine and its metabolite morphine in ultra-rapid metabolizers due to CYP2D6 duplication. Published in 2007. Available from: The Pharmacogenomics Journal, 7, 257-265.
- S Clarke et al. Naloxone in opioid poisoning: walking the tightrope. Published in 2005. Available from: Emergency Medicine Journal, 22, 612-616.
- Gov.uk. Widening the availability of naloxone. Published in 2019. Available from: [LINK].
- E Schiller et al. Opioid Overdose. Published in 2020. Available from: [LINK].
- Figure 1. Thomas Bonini. Miosis caused by opioid consumption. License: [Public Domain]. Available from: [LINK]




