- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
Loss of vision can be caused by ophthalmological (due to eye pathology) and neurological (due to nervous system pathology) conditions. This article will cover neurological causes of visual loss.
Loss of vision can have a significant impact on quality of life, mental health, and wellbeing, as well as being an independent risk factor for falls.1
The Royal National Institute of Blind People (RNIB) estimated an incidence of 0.14% of the UK population experienced new sight loss or blindness in 2016-17.
An extrapolated lifetime risk of all cause of sight loss (including treatable causes) was estimated at 36.3% in the UK. This means one in three people are at risk of sight loss requiring intervention or treatment during their lifetime.2
Stratifying patients with visual loss into life-threatening, sight-threatening and non-emergency is a vital skill. In addition, knowing if the visual loss is ophthalmological or neurological is important to know the correct referral pathway.
For more information on ophthalmological causes of sudden loss of vision, see the Geeky Medics guide to sudden painless loss of vision.
Aetiology
Visual loss can be caused by pathophysiological processes throughout the visual pathway (Figure 1, Table 1).
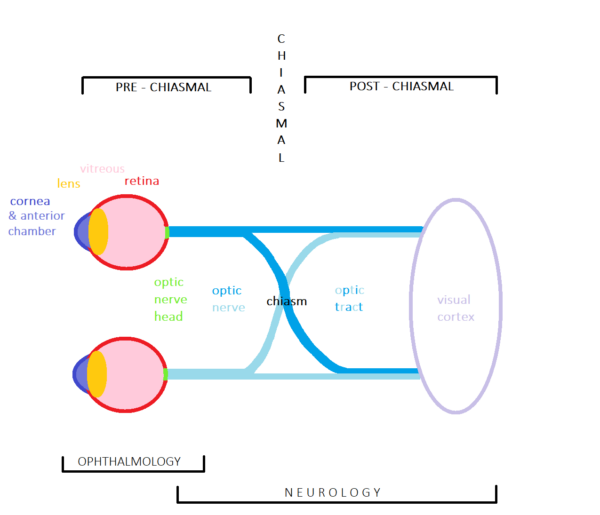
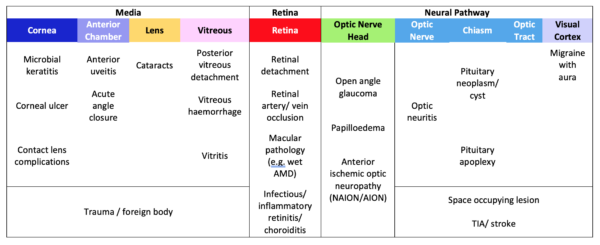
Neurological visual loss results from nervous system pathology which occurs anywhere along the visual pathway, from the optic nerve to the visual cortex in the occipital lobe of the brain. Anatomically, these are termed retrobulbar, as they occur posterior to the globes.
The most common neuro-ophthalmological symptoms include altered colour perception, diplopia, loss of vision and characteristic visual field defects. This article will focus on conditions where vision loss is the principal symptom.
Establishing the time course of symptoms along with a focused history can be a valuable indicator as to what pathological process is occurring.
This will help guide optimal management aiming to avoid any permanent visual deficit.
- Sudden onset of neurological visual loss is often vascular in aetiology and includes cerebrovascular disease (TIA and stroke).
- Subacute onset is more suggestive of infective or inflammatory causes, such as optic neuritis.
- Gradual or worsening vision loss is more consistent with a degenerative pathology or a space-occupying lesion.
- Transient and intermittent symptoms can be seen in various neurological conditions, including migraine with aura, amaurosis fugax, TIA and optic neuritis.
Optic neuritis
Optic neuritis is an inflammatory condition affecting any part of the optic nerve, causing acute or sub-acute painful and, usually, monocular vision loss.
It is the presenting feature of multiple sclerosis (MS) in 15-20% of patients and occurs at some stage in at least half of patients diagnosed with MS.3
Aetiology
Optic neuritis generally refers to acute demyelinating optic neuritis which is highly associated with MS but can also be idiopathic, in which case it is described as a clinically isolated syndrome (CIS).
Rarely, optic neuritis can result from systemic disorders. These include infections such as tuberculosis and syphilis and autoimmune/inflammatory conditions such as systemic lupus erythematosus and sarcoidosis.4
Risk factors
Optic neuritis is most common in women between 18-50 years old and in Caucasian populations.
Clinical features
Typical symptoms of demyelinating optic neuritis include:
- Eye pain that is characteristically worse on eye movement
- Monocular vision loss developing over hours to days; this varies from a slight blur to no light perception
- Impaired colour vision in the affected eye, typically red desaturation
Typical clinical examination findings of demyelinating optic neuritis include:
- Reduced visual acuity
- Relative afferent pupillary defect (RAPD) on the affected side
- Visual field defects: commonly central but can be any pattern
Only 33% of patients have overt optic disc swelling (papillitis) with hyperaemia. The remaining 66% have retrobulbar optic neuritis, with a normal fundoscopic examination (Figure 2).5
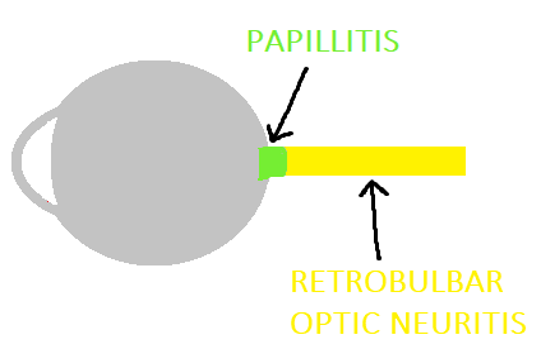
Investigations
Optic neuritis is a clinical diagnosis. However, MRI of the brain and orbits is usually requested to look for other areas suggestive of demyelination which could indicate a diagnosis of MS.
Management
Vision starts to recover within a month without treatment. There is no effect on long-term visual outcome with medical treatment, however, systemic corticosteroids have been shown to hasten visual recovery.6
Patients should be referred to neurology if MS is suspected.
Complications
Following recovery and despite the return of normal visual acuity, clinical findings such as RAPD, red desaturation, and optic nerve pallor (atrophy) commonly persist (Figure 4).
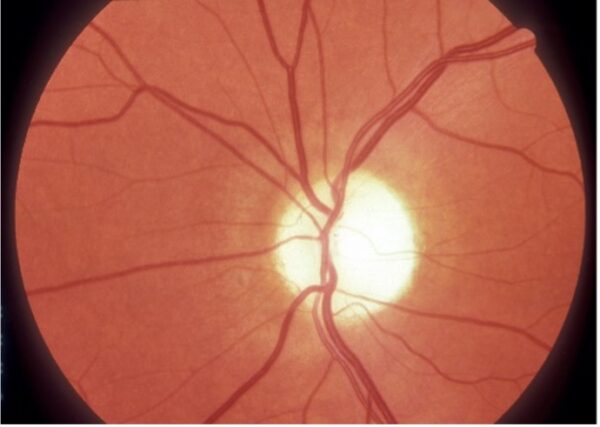
Key points
- Optic neuritis usually refers to acute demyelinating optic neuritis which is highly associated with MS.
- Optic neuritis typically presents with monocular symptoms including pain on eye movement, variable vision loss occurring over hours to days and impaired colour vision
- Symptoms gradually resolve over several weeks without treatment
Migraine with aura
Migraine with aura is the most common cause of transient vision loss in young adults.
Aetiology
Migraine is a complex condition, and the pathophysiology is not fully understood. It can be precipitated by a trigger, with common examples include hormonal changes and dysregulation of sleep and meals.
Risk factors
There is a strong genetic predisposition to developing migraine with aura.
Other common risk factors proposed include female sex, stress, obesity, diabetes, and head injury.
Clinical features
Typical symptoms of migraine with aura include:
- Evolving positive visual phenomenon such as flickering, scintillating, or bright geometric shapes, that completely resolve. Often described as bilateral and present when eyes are closed, typically lasting 10-60 minutes.
- Can be associated with or followed by a headache.
- An aura of atypical duration or accompanied by negative features, such as hemianopia, are rarer.
For more information, see the Geeky Medics guide to headache history taking.
Clinical examination of migraine with aura (including visual field testing) should be unremarkable.
Investigations
Migraine with aura is a clinical diagnosis and does not often require further investigation.
If there is any unexplained focal neurological deficit found on examination, further investigation should be considered, such as serum inflammatory markers, carotid duplex ultrasound, ophthalmological examination, or neuroimaging.
Management
Acute medical therapy is taken at the onset of migraine symptoms. These include analgesics such as high dose aspirin, ibuprofen or triptans.
Primary prevention aims to manage the frequency of migraines. Avoidance of triggers and lifestyle modification are included here along with medical prophylaxis, such as propranolol or amitriptyline, which are considered when there is a significant reduction in quality of life associated with migraines.
The combined oral contraceptive pill should be discontinued in patients with migraine with aura due to the small increased risk of ischaemic stroke.8
Complications
Rarely infarction or seizure can occur along with prolonged symptoms, which include status migrainosus or persistent aura without infarction.9
Key points
- Migraine with aura is the most common cause of transient vision loss in young adults.
- It typically presents as evolving visual phenomenon that demonstrates complete reversibility, with phenomena most commonly lasting 10-60 minutes.
- It is managed acutely with NSAIDs or triptans, and in some cases with medical prophylaxis (e.g. propranolol).
- The combined oral contraceptive pill should be discontinued due to the small increased risk of ischaemic stroke.
Amaurosis fugax
Amaurosis fugax translates as “fleeting darkness” and can be either monocular or binocular. It is considered a form of transient ischaemic attack (TIA).
Almost all patients will present once the episode has resolved. An accurate history detailing the timing of events and characteristics of vision loss is crucial.
Aetiology
Amaurosis fugax describes a transient monocular vision loss which is a result of ischaemia to the retina, choroid, or optic nerve from an arterial embolus, usually in the retinal artery from ipsilateral carotid artery disease (Figure 5).10
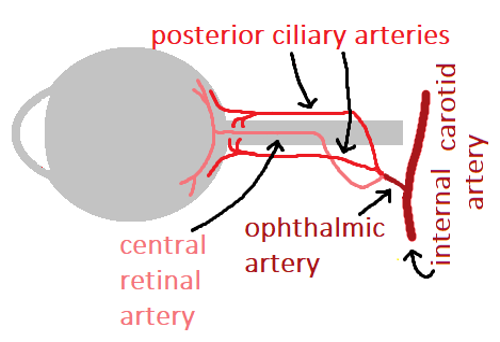
Risk factors
Amaurosis fugax usually occurs in patients over the age of 50 with vascular risk factors, such as hypertension, hypercholesterolaemia, smoking and previous history of TIA/stroke (cerebrovascular disease).
Clinical features
The typical presentation of amaurosis fugax is transient vision loss in one or both eyes occurring abruptly, reaching maximum severity within seconds and lasting seconds to minutes, followed by full visual recovery.
It is most commonly a negative visual phenomenon, described as a blackout, or ‘greying out’ of vision.
An ophthalmological examination will typically be normal.
Investigations
Relevant investigations include:
- Inflammatory markers: routinely tested as temporal/giant cell arteritis must be excluded
- Carotid artery imaging: to assess for carotid artery stenosis
- Cardiac investigations: looking for atrial fibrillation or other stroke risk factors
Neuroimaging is not indicated unless there is clinical suspicion of an alternative diagnosis that would be detected with CT or MRI scanning.
Management
Patients presenting acutely are offered 300mg of aspirin unless contraindicated. Urgent referral to a specialist TIA clinic within 24 hours is required.
Once the diagnosis is confirmed, secondary prevention is initiated (e.g. statin therapy, antiplatelet therapy and optimising blood pressure control).
Complications
The risk of ischaemic stroke in patients with amaurosis fugax is estimated to be around 2% per year. This increases to 8.4% in the presence of significant ipsilateral carotid artery stenosis.11
Key points
- Amaurosis fugax is a form of TIA, usually from an arterial embolus arising from carotid artery disease.
- Transient vision loss occurs in one or both eyes lasting seconds to minutes followed by full recovery.
- Acute management often involves 300mg aspirin, unless contraindicated.
- Urgent referral to a specialist TIA clinic is required who will formally diagnose, assess risk and manage risk factors.
- The risk of ischaemic stroke following amaurosis fugax is around 2% per year.
Pituitary adenoma
Pituitary adenomas account for up to 15% of symptomatic intracranial neoplasms and are the most common cause of optic chiasm compression in adults.
Other pathologies that affect the sellar space include cysts, vascular events (apoplexy), inflammation, infection, and metastases.
Aetiology
The adenoma compresses the optic chiasm leading to vision loss. The most common pituitary pathology presenting with vision loss is a non-functioning pituitary macroadenoma.12
Risk factors
Pituitary adenomas occur predominantly in adulthood.
3% of cases are associated with multiple endocrine neoplasia (MEN type 1) however, the majority occur in isolation. Genetic factors have been shown to contribute to risk.
Clinical features
Typical symptoms of pituitary adenoma include:12
- Headache: the most common presenting symptom
- Vision changes: loss of peripheral vision is often so insidious that many patients remain asymptomatic with or without the presence of a visual field defect
- Symptoms of pituitary dysfunction: including weight gain, mood changes/irritability, loss of libido, dizziness, erectile dysfunction and infertility
Typical clinical examinations findings of pituitary adenoma include:12
- Reduced visual acuity in advanced chiasmal compression
- Signs of pituitary dysfunction: such as gynecomastia and acromegaly
- Visual field loss: classically bitemporal hemianopia (Figure 6)
Investigations
If a visual field defect is present, an urgent endocrine assessment is required where a firm biochemical diagnosis will be established. A pituitary MRI will then be arranged by secondary care if deemed necessary.
Management
Management varies depending on the lesion, but if the visual field is affected, transsphenoidal surgery offers the best chance of restoring vision.13
Complications
Pituitary apoplexy is a life-threatening complication, where sudden haemorrhage or infarction of the tumour occurs. It presents with severe headache, nausea, loss of vision and reduced consciousness.
Corticosteroids must be started immediately followed by confirmation of diagnosis by neuroimaging. Surgical decompression may also be indicated.
Key points
- Pituitary adenoma is the most common cause of optic chiasm compression in adults and can present with vision loss
- Headache and bitemporal hemianopia are the most common clinical features, however many patients are asymptomatic
- Urgent endocrine assessment is required to establish a biochemical diagnosis, often followed by a pituitary MRI
- Transsphenoidal surgery offers the best chance at restoring vision
- Pituitary apoplexy is a life-threatening vascular event that can occur
Ischaemic stroke
Any intracranial lesion along the visual pathway can cause vision loss. Vascular causes are the most common in adults, followed by traumatic brain injury and brain tumours.
This section will discuss a new presentation of homonymous hemianopia as a result of ischaemic stroke. The FAST (face, arm, speech, time) screening tool may miss this presentation.
Aetiology
Homonymous hemianopia occurs due to a unilateral lesion posterior to the chiasm (retro-chiasmal), which affects the contralateral visual fields of each eye.
Homonymous hemianopia presents in both anterior and posterior circulation stroke syndromes, under the Oxford stroke classification.
Isolated homonymous hemianopia can be classified as a posterior circulation stroke (POCS).
Risk factors
Ischaemic stroke usually occurs in patients over the age of 50 with vascular risk factors, such as hypertension, hypercholesterolaemia, smoking and previous history of cerebrovascular disease.
Over 40% of POCS are attributed to cardioembolism (e.g. atrial fibrillation).
Clinical features
Patients usually describe monocular vision loss or difficulty with certain tasks such as reading. Visual acuity is not usually affected.
Other acute neurological deficits can occur, some of which are described below.
Clinical examination findings vary depending on the anatomical location:
- Optic tract lesions will present with homonymous hemianopia and ipsilateral RAPD with temporal vision loss, optic nerve atrophy (“optic tract syndrome”) ± hemiparesis
- Optic radiations lesions will present as incongruous (asymmetric) homonymous hemianopia ± aphasia, memory deficits, seizures, and hallucinations
- Occipital cortex lesions will present as isolated homonymous hemianopia that is highly congruous (equal) ± macular sparing
- Brainstem lesions present with any combination of homonymous hemianopia, cranial nerve palsy, nystagmus, vertigo, dysarthria, and ataxia

Investigations
Rapidly developing neurological symptoms should immediately raise suspicion of an acute neurovascular event, with the presence of isolated homonymous hemianopia raising the index of suspicion to a posterior circulation stroke.
An urgent non-contrast CT scan should be performed to exclude intracranial haemorrhage. CT is less sensitive in detecting posterior circulation (60%) compared to anterior circulation (>90%) strokes.14
MRI offers a higher sensitivity along with being able to distinguish mass from vascular aetiologies.15 MRI with DWI sequencing within 12 hours of the event is highly sensitive (92%) in identifying acute ischaemic stroke.16
Identification of underlying general and cardiac risk factors (e.g. ECG, bloods, carotid dopplers, echocardiogram) should also be carried out.
Management
Acute stroke management includes resuscitation and direct admission to a specialist stroke unit after an initial assessment.
If within 4.5 hours of the onset of symptoms, with no contraindications, thrombolysis may be offered.
If thrombolysis is contraindicated or not available, 300mg aspirin orally along with a proton pump inhibitor must be offered within 24 hours when haemorrhage is excluded unless also contraindicated.14
In some UK centres, thrombectomy is available as an acute treatment of ischaemic stroke.
Stroke prevention strategies are key to preventing recurrence and aim to control modifiable risk factors.15
Advice to cease driving is crucial, however, up to 50% of visual field defects recover spontaneously within the first month, so observation is important.17
Referral to low vision services for rehabilitation assessment can be beneficial if the field defect remains, as visual recovery after six months is rare.
In the United Kingdom, patients must be advised to inform the Driver and Vehicle Licensing Agency (DVLA).
Complications
An important complication to consider is a further stroke or vascular cerebral event.
Key points
- Homonymous hemianopia occurring in isolation is classified as an isolated posterior circulation stroke syndrome (POCS) and is a rare cause of vision loss
- Other neurological deficits can occur alongside homonymous hemianopia which aids identification of the pathological location along the visual pathway
- A suspected acute stroke must be referred directly to a specialist stroke unit for management and prevention strategy implementation
- Over 50% of visual field defects recover spontaneously within the first month; recovery after six months is rare
Final summary
If a patient’s vision loss is suspected to be a result of a life-threatening or sight-threatening condition, they must be referred urgently. Importantly, assessing whether vision loss is ophthalmological or neurological in nature will allow the most appropriate referral and onward management.
Reviewer
Dr Kathryn A W Knight
Neurology registrar
NHS Tayside
Editor
Dr Chris Jefferies
References
- Jack, CIA. Prevalence of Low Vision in Elderly Patients Admitted to an Acute Geriatric Unit in Liverpool: Elderly People Who Fall Are More Likely to Have Low Vision. Published in 1995. Available from: [LINK]
- Department of Health. National Service Framework for Older People. Available from: [LINK]
- Balcer, LJ. Clinical practice. Optic neuritis. Published in 2006. Available from: [LINK]
- Abel, A. Critical review: Typical and atypical optic neuritis. Published in 2019. Available from: [LINK]
- Eyewiki. Demyelinating Optic Neuritis. Updated in 2021. Available from: [LINK]
- Beck, RW. The Course of Visual Recovery after Optic Neuritis: Experience of the Optic Neuritis Treatment Trial. Published in 2020. Available from: [LINK]
- Clare Gilbert. International Centre for Eye Health, London School of Hygiene & Tropical Medicine. Optic atrophy. License: [CC BY-SA]
- Allais, G. Oral contraceptives in migraine. Published in 2009. Available from: [LINK]
- Alyssa E. Blumenfeld. Complicated Migraines. Published in 2016. Available from: [LINK]
- Pia Kvickstrom. Amaurosis fugax: risk factors and prevalence of significant carotid stenosis. Published in 2016. Available from: [LINK]
- De Schryver ELLM. Type of stroke after transient monocular blindness or retinal infarction of presumed arterial origin. Journal of neurology, neurosurgery and psychiatry. Published in 2006. Available from: [LINK]
- UpToDate. Causes, presentation, and evaluation of sellar masses. Available from: [LINK]
- Kerrison, JB. Stages of improvement in visual fields after pituitary tumor resection. Published in 2000. Available from: [LINK]
- NICE. Stroke and transient ischaemic attack in over 16s: diagnosis and initial management. Available from: [LINK]
- Aine Merwick and David Werring. Posterior circulation ischaemic stroke. BMJ : British Medical Journal. Published in 2014. Available from: [LINK]
- Haz-Edine Assemlal. Recent advances in diffusion MRI modeling: Angular and radial reconstruction. Published in 2011. Available from: [LINK]
- Christopher S. Gray. Recovery of visual fields in acute stroke: Homonymous hemianopia associated with adverse prognosis. Published in 1989. Available from: [LINK]




