- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
This article aims to provide an overview of the fundoscopic appearances of common retinal pathologies which may be asked to identify in an OSCE scenario.
You may also be interested in our guide to fundoscopy.
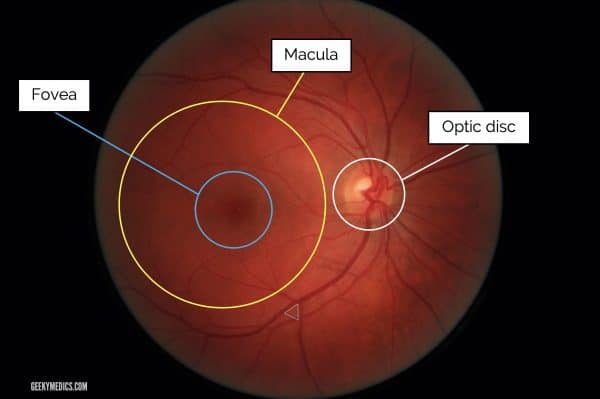
Normal appearance of the retina
The key parts of the retina to recognise are the optic nerve head (optic disc) and the macula.
The optic nerve is found by tracing any of the blood vessels to the point of coalescence (branching vessels form an arrow pointing towards the disc, as shown below).
The macula is found lateral (temporal) to the optic nerve head. The central part of the macula, the “fovea” is about the same diameter as the optic disc and appears darker than the rest of the macula due to the presence of an additional pigment.
Diabetic retinopathy
Diabetic retinopathy is a form of micro-angiopathy causing damage to the small blood vessels of the retina as a result of hyperglycaemia.
The most important risk factors for developing diabetic retinopathy are the duration of diabetes and poor diabetic control. Other vascular risk factors like hypertension, hyperlipidaemia and smoking also contribute to the risk of developing diabetic retinopathy.
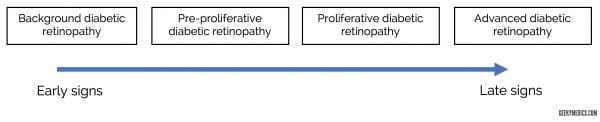
The spectrum of diabetic retinopathy is shown below. Many medical schools focus on the grading system below, though others do exist.
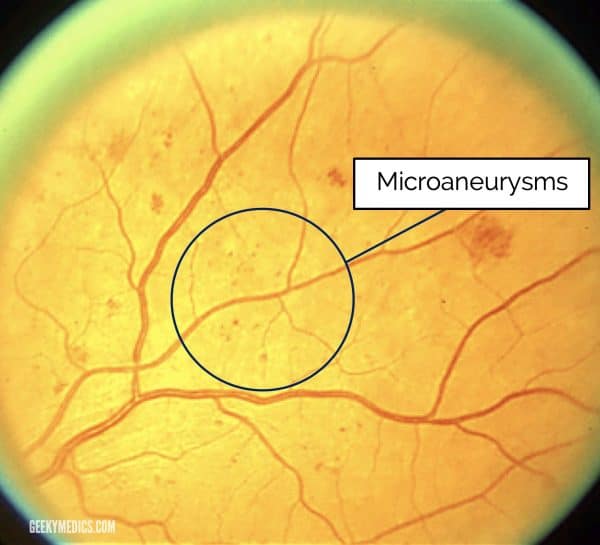
Background diabetic retinopathy
Microaneurysms
Microaneurysms are localised outpouchings of capillaries that leak plasma constituents into the retina. They may be clinically indistinguishable from small dot and blot haemorrhages (see below).
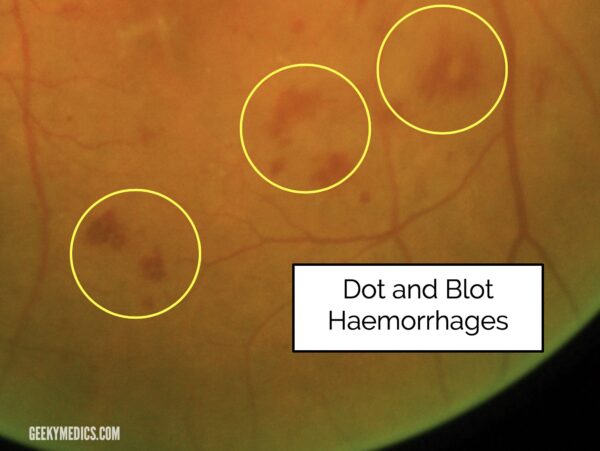
Dot and blot haemorrhages
Dot and blot haemorrhages arise from bleeding capillaries in the middle layers of the retina.
They may look like microaneurysms if small enough. It is not particularly important to be able to distinguish between small haemorrhages and microaneurysms as they are both parts of pre-proliferative retinopathy.
Pre-proliferative diabetic retinopathy
The presence of retinal ischaemia represents a progression from background diabetic retinopathy to the pre-proliferative stage.
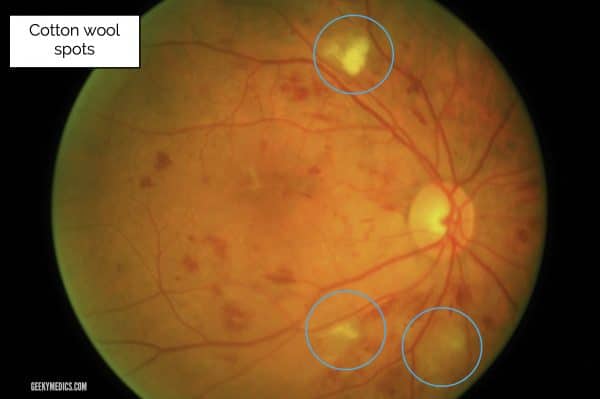
Cotton wool spots
Cotton wool spots appear as small, fluffy, whitish superficial lesions.
They are accumulations of dead nerve cells from ischaemic damage.
Other signs
Other signs of pre-proliferative retinopathy include venous changes and intraretinal microvascular anomalies (IRMA) but you would not be expected to know or recognise them at the undergraduate level.
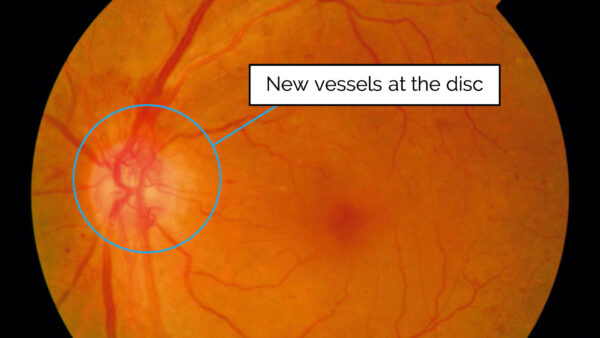
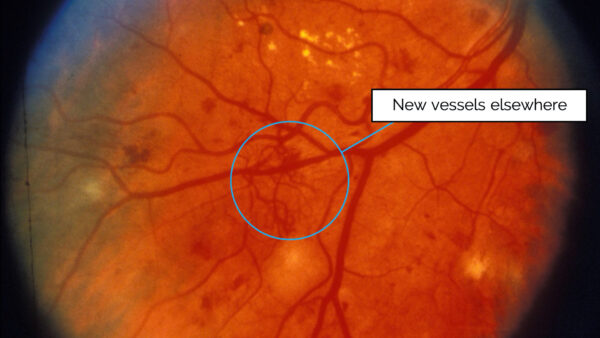
Proliferative diabetic retinopathy
Insufficient retinal perfusion results in the production of vascular endothelial growth factor (VEGF) which results in the development of new vessels on the retina (neovascularisation).
These new vessels may either be at the disc, termed “new vessels at the disc” (NVD), or over the other areas of the retina “new vessels elsewhere” (NVE).
Advanced diabetic retinopathy
Advanced diabetic retinopathy results in:
- Recurrent vitreous haemorrhage from bleeding areas of neovascularisation
- Tractional retinal detachments as areas of neovascularisation grow into the vitreous and form fibrous bands suspending the retina
- Rubeosis as neovascularisation occurs at the iris and drainage angle resulting in increased intraocular pressure and progressive glaucoma
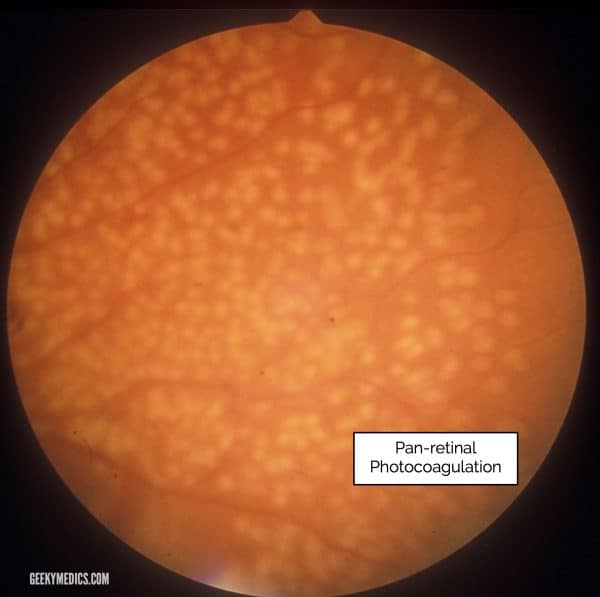
Pan-retinal photocoagulation (PRP)
Pan-retinal photocoagulation is the primary treatment for proliferative diabetic retinopathy.
The rationale behind the treatment is to reduce the production of VEGF by reducing the oxygen demand from the peripheral retina.
Clinically it is seen as clusters of burn marks on the retina which have been created by the laser used in the treatment process.
Diabetic maculopathy
Diabetic maculopathy refers to the presence of exudates and/or macular oedema at the macula.
You are unlikely to be asked to identify oedema as it is hard to visualise using a direct ophthalmoscope.
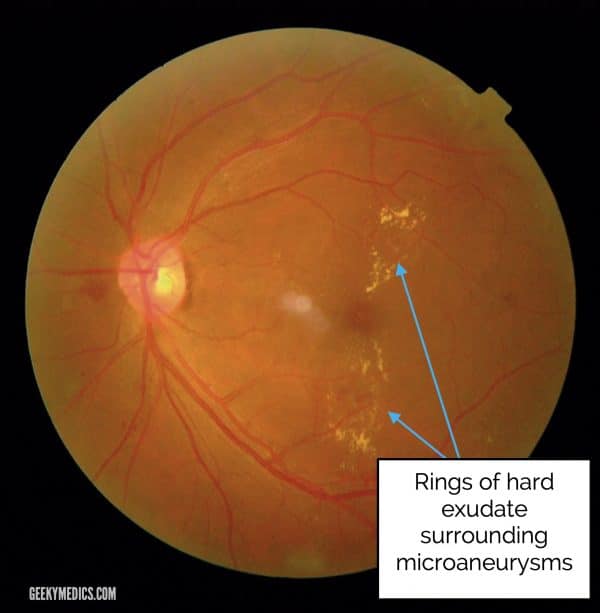
Hard exudates
Hard exudates are waxy yellow lesions with relatively distinct margins arranged in clumps or rings, often surrounding leaking microaneurysms.
They are called “hard exudates” to distinguish them from “soft exudates”, an older term for cotton wool spots (see below).
Hypertensive retinopathy
Chronic hypertension
Hypertensive retinopathy is graded as follows:
- Grade 1 disease: the changes of early hypertensive retinopathy are subtle, with generalised arteriolar narrowing.
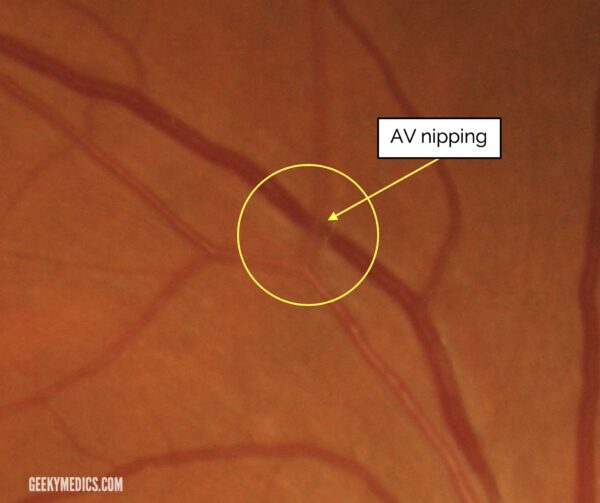
- Grade 2 disease: development of areas of focal narrowing, and compression of venules at sites of arteriovenous crossing (AV nipping).
- Grade 3 disease: development of features similar to those of diabetic retinopathy, namely retinal haemorrhages, hard exudates and cotton wool spots.
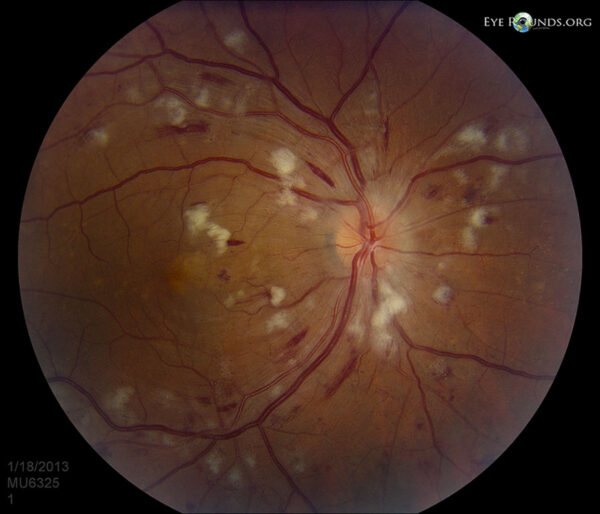
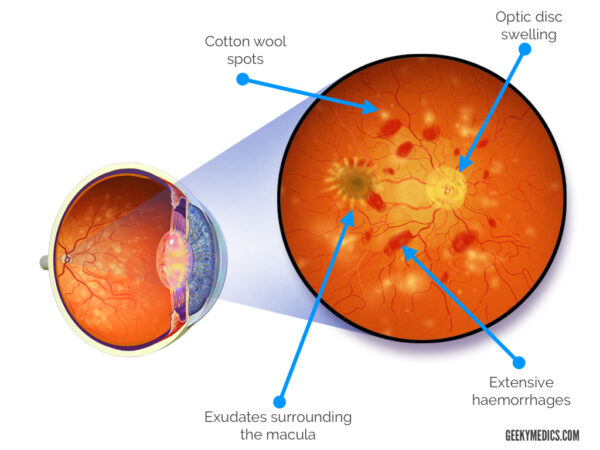
Malignant hypertension
Malignant hypertension typically presents with grade 4 hypertensive eye disease, which includes all the features of grade 3, with the addition of optic disc swelling. Other features include headaches, eye pain, reduced visual acuity and focal neurological deficits.
Initial management typically involves antihypertensives and emergency hospital admission.
The image below shows extensive haemorrhages, cotton wool spots, optic disc swelling and a ring of exudates around the macula (macular star).
See our guide to hypertension for more details.
Optic nerve pathologies
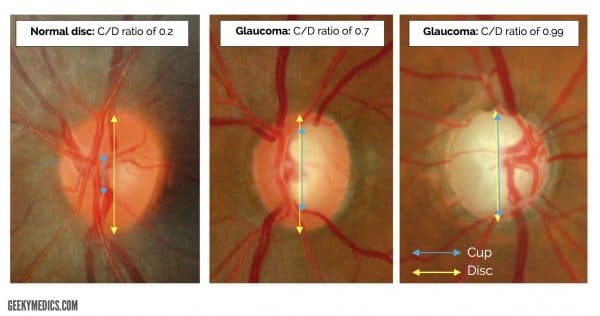
Glaucomatous disc changes
Glaucoma is defined as a progressive optic neuropathy that is associated with visual field loss and usually raised intraocular pressure (IOP).
Glaucoma results in irreversible loss of nerve fibres, leading to an appearance of “cupping” or an increased vertical cup-to-disc ratio (C/D ratio).
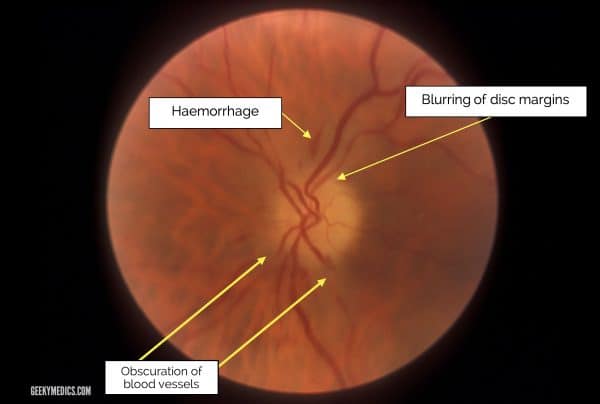
Papilloedema/optic nerve swelling
Papilloedema refers to optic disc swelling secondary to raised intracranial pressure.
“Optic disc swelling” is a general term referring to any cause of a swollen optic disc.
Causes of optic disc swelling include intracranial space-occupying lesions, optic neuritis (e.g. multiple sclerosis), malignant hypertension and uveitis (e.g. tuberculosis, sarcoidosis).
Optic disc margins appear blurred and small haemorrhages may be noted.
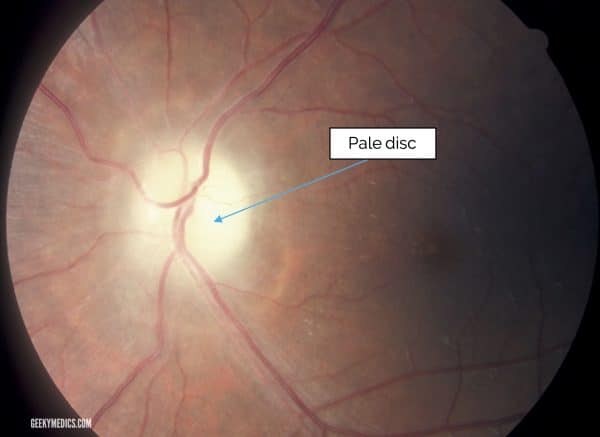
Optic nerve atrophy
Optic nerve atrophy involves the death of nerve fibres within the optic nerve. This results in a pale optic disc as opposed to the usual pink appearance.
Primary optic nerve atrophy is caused by inflammation (optic neuritis), glaucoma or general retinal ischaemia.
Secondary optic nerve atrophy is caused by longstanding papilloedema.
Investigation of this condition often requires neuroimaging to rule out life-threatening intracranial causes.
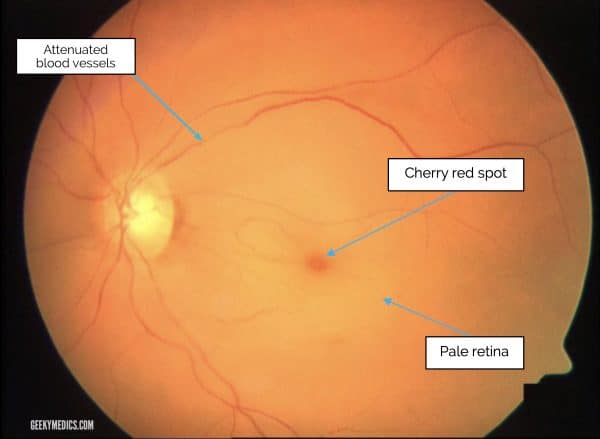
Vascular occlusive diseases
Central retinal artery occlusion (CRAO)
Central retinal artery occlusion presents with sudden, profound loss of vision.
It is most frequently caused by emboli obstructing the retinal artery (e.g. stroke). Occasionally, it can be caused by vasculitis (e.g. giant cell arteritis).
Classical appearance is of a “cherry-red spot”. This occurs due to the intact reflex of the fovea standing out against a pale, ischaemic retina.
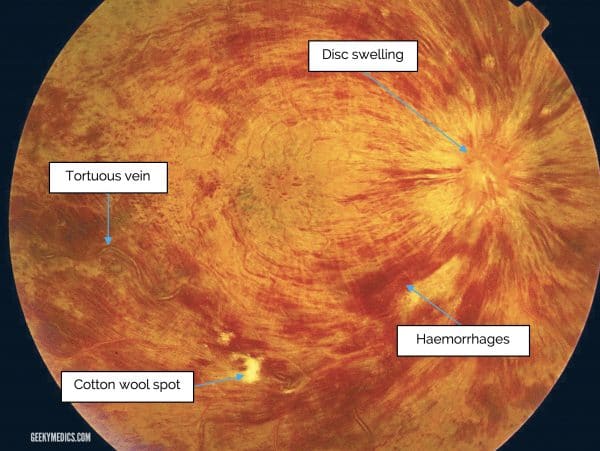
Central retinal vein occlusion (CRVO)
Central retinal vein occlusion occurs secondary to atherosclerotic thickening of the central retinal artery compressing the central retinal vein at a common crossing point.
Occlusion of the vein results in retinal hypoxia, and the resultant endothelial cell damage leads to extravasation of blood.
Fundoscopy typically reveals severe tortuosity, engorgement of retinal veins, deep haemorrhages, cotton wool spots and optic disc swelling.
Macular pathology
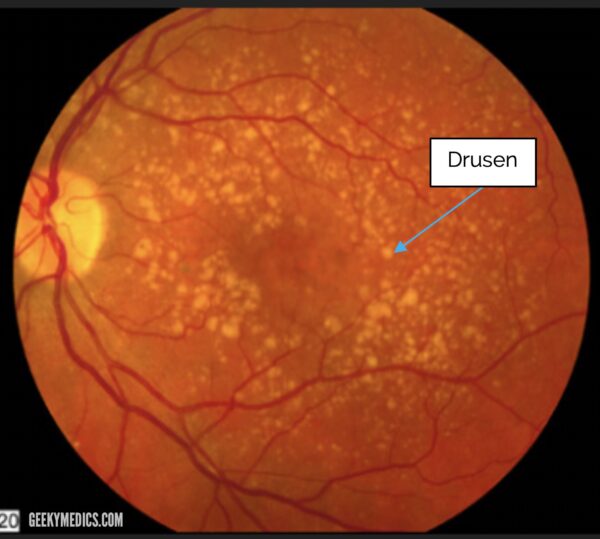
Age-related macular degeneration (AMD)
Age-related macular degeneration is a degenerative disorder of the macula and the most common cause of irreversible blindness in the elderly population.
The most common form of AMD (90% of cases) is the “dry” variant, termed so as it produces no exudation or neovascularisation.
Early signs of AMD include the presence of yellow-white deposits which are termed “drusen”.
References
Reference images
- Adapted by Geeky Medics. Normal fundus photograph provided courtesy of Yandle on Flickr for re-use under CC BY 2.0.
- Adapted by Geeky Medics. All diabetic retinopathy, optic nerve pathologies, and CRVO photographs provided by Community Eye Health on Flickr for non-commercial reuse under CC BY-NC 2.0.
- Adapted by Geeky Medics. Photograph of AV nipping originally provided by Frank Wood for re-use under CC BY 3.0.
- Adapted by Geeky Medics. Photograph of a cherry-red spot originally provided by Fieß et al. and BioMed Central Ltd. Under CC BY 2.0.
- Adapted by Geeky Medics. Photograph of AMD provided by National Eye Institute on Flickr for re-use under CC BY 2.0.
- Adapted by Geeky Medics. Blausen.com staff (2014). Medical gallery of Blausen Medical 2014. Licence: [CC BY 3.0 ].
- Adapted by Geeky Medics. Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a CC-ND-NC 3.0.
Reference texts
- Kanski JJ, Bowling B. Clinical Ophthalmology: A Systematic Approach. Edinburgh, Elsevier/Saunders; 2015
- Vaughan CJ, Delanty N. Hypertensive emergencies. Lancet. 2000; 356:411–417
Reviewer
Dr Ashley Simpson
Ophthalmology Registrar