- 📖 Geeky Medics OSCE Book
- ⚡ Geeky Medics Bundles
- ✨ 1300+ OSCE Stations
- ✅ OSCE Checklist PDF Booklet
- 🧠 UKMLA AKT Question Bank
- 💊 PSA Question Bank
- 💉 Clinical Skills App
- 🗂️ Flashcard Collections | OSCE, Medicine, Surgery, Anatomy
- 💬 SCA Cases for MRCGP
To be the first to know about our latest videos subscribe to our YouTube channel 🙌
Introduction
Pulmonary embolism (PE) is the luminal obstruction of one or more pulmonary arteries by an embolised venous thrombus, but can also be due to an embolised solid, liquid, or gas.1
Venous thromboembolism (VTE) is an umbrella term encompassing both pulmonary embolism (PE) and deep vein thrombosis (DVT).1
In the United Kingdom (UK), PE and DVT together make up the third most common cardiovascular disease (after acute myocardial infarction and stroke). PE has an overall annual incidence of 53-162 per 100,000 people. Nearly 70,000 hospital episodes of PE were reported between 2021-2022, resulting in over 35,000 admissions.2
Aetiology
A thrombus refers to a blood clot that is formed within a blood vessel and remains in its place of origin. Embolism describes the process where the bloodstream carries a detached intravascular solid, liquid, or gaseous mass from its origin to a distant site.1
Most PEs originate as thrombi due to deep vein thrombosis, most frequently in the calf veins.3
Other causes include:3
- Fat embolism
- Air embolism
- Amniotic fluid embolism
- Septic emboli
- De novo thrombosis (rare)
Pathophysiology
In a PE, an embolus detaches from its origin and embolises to the pulmonary arteries through the systemic venous system and the right heart, finally causing luminal obstruction of one or more pulmonary arteries.3,5
Pathological consequences of pulmonary embolism:3,5
- Ventilation/perfusion mismatch → impaired gaseous exchange → hypoxaemia (type 1 respiratory failure) and tachypnoea
- Pulmonary arterial hypertension (due to increased pulmonary vasculature resistance) → right ventricular overload +/- dysfunction
- Pleural and lung inflammation and infarction → pleuritic chest pain +/- haemoptysis
The diagnosis of PE can be challenging, and these pathological changes do not always manifest with the above clinical features.
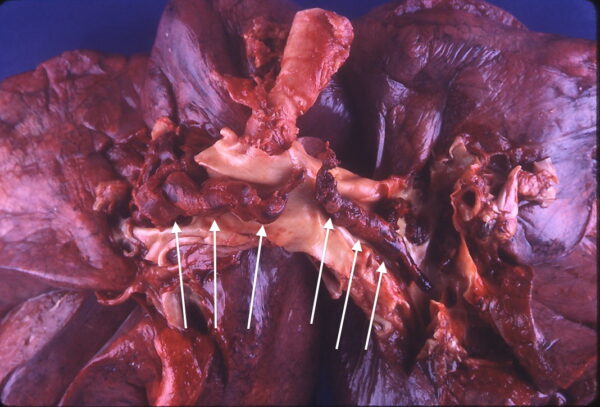
Risk factors
Risk factors for PE include and are not limited to:3,4,5
- Diagnosis of DVT: ~45-50% of people with PE have concurrent DVT
- Increasing age
- Previous VTE
The Virchow’s triad for VTE:3,4,5
- Venous stasis: prolonged immobilisation (e.g. bed rest >5 days, major surgery within the last 2 months, recent trauma or fracture, paralysis of the lower limb, long-haul flights), venous insufficiency of the lower limb.
- Hypercoagulable state: active malignancy, pregnancy and postnatal period, thrombophilia (e.g. antiphospholipid syndrome, factor V Leiden mutation), use of combined hormonal contraception and oral hormone replacement therapy.
- Endothelial injury: trauma, surgery, venous harvest, cigarette smoking, obesity.
Concurrent DVT, prolonged immobilisation and previous VTE events are strong risk factors for PE (they form part of the 2-level PE Wells score).
Clinical features
The signs and symptoms of pulmonary embolism are highly non-specific, making PE challenging to diagnose.
History
Typical symptoms of PE may include:2,3,4,9
- Dyspnoea: the most common feature
- Tachypnoea (~20-40% cases)
- Pleuritic chest pain (~40% of cases)
- Features of concurrent DVT (typically unilateral red, painful swollen leg)
- Haemoptysis
Other possible symptoms include:2,3,4,9
- Retrosternal chest pain (due to right ventricular ischaemia)
- Cough
Clinical examination
Patients with a suspected PE require a comprehensive respiratory and cardiovascular examination. Acutely unwell patients will require an ABCDE approach.
Typical clinical findings on examination may include:2,3,4,9
- Tachycardia
- Tachypnoea
- Hypoxia
- Low-grade fever
- Pleural rub
- Gallop rhythm, a wide split-second heart sound and tricuspid regurgitant murmur.
Massive pulmonary embolism
Features of a massive PE include:2,3,4,9
- Haemodynamic instability: hypotension and cardiogenic shock
- Presyncope/syncope
- Elevated jugular venous pressure (JVP)
Investigations
Bedside investigations
A 12-lead ECG should always be performed in the context of chest pain. In the context of PE, it should be used to exclude possible differential diagnoses (e,g. acute coronary syndrome). ECG features alone are not diagnostic of PE.
Possible ECG findings include:4,11
- Sinus tachycardia: the most common finding
- Right ventricular strain pattern: T wave inversion in anterior leads (V1-V4) +/- inferior leads (II, III, aVF)
- Right bundle branch block (RBBB)
- Right axis deviation (RAD)
- The classic ‘S1Q3T3’ ECG change is only seen in <20% patients – large S wave in lead I, large Q wave in lead III, and inverted T wave in lead III
Laboratory investigations
Relevant laboratory investigations include:2,3
- D-dimer levels: often raised, typically >500 ng/mL (see below)
- Full blood count (FBC): may demonstrate leucocytosis due to acute inflammation but is non-specific
- Urea and electrolytes (U&E): often unremarkable, baseline renal function results can be used to guide investigations and treatment
- Liver function tests (LFTs): often unremarkable, they can be used to guide treatment
- Cardiac biomarkers (troponin and BNP) are not used in diagnosing PE but can provide prognostic information.
- Coagulation studies: should be established before starting treatment and can guide treatment.
D-dimer
D-dimer test has a high sensitivity but low specificity for VTE. Many conditions can result in an elevated D-dimer in the absence of VTE:2,13
- Pregnancy
- Malignancy
- Liver disease
- Severe infection or inflammatory disease
- Disseminated intravascular coagulation (DIC)
- Recent trauma/surgery/hospitalised patients
In these patients, D-dimer testing has no value in diagnosing VTE.
Imaging
Chest X-ray
Chest X-ray should be used to exclude possible differential diagnoses (e.g. pneumothorax and pneumonia). Most patients with PE have a normal chest X-ray.10
Possible chest X-ray findings in PE include:10
- Wedge-shaped pulmonary infarction: wedge-shaped opacification without air bronchograms
- Atelectasis
- Pleural effusion
- Raised hemidiaphragm
CTPA (CT pulmonary angiogram)
CTPA is the preferred investigation for the definitive diagnosis of PE.
Diagnosis of PE is confirmed by direct visualisation of a thrombus within a pulmonary artery lumen.
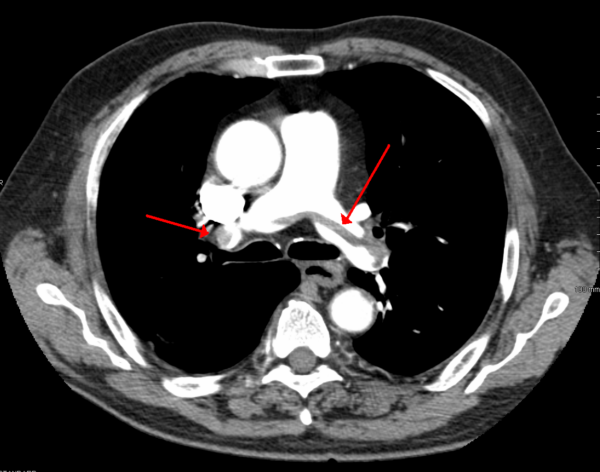
Alternative to CTPA in the diagnosis of PE
NICE recommends ventilation-perfusion (V/Q) planar scan or SPECT as the investigation of choice instead of CTPA in the following patients:12
- Severe renal impairment (eGFR < 30 mL/min/1.73m2)
- Allergic to contrast media
- At high risk from irradiation (e.g. pregnancy)
Both V/Q planar scan and SPECT have a lower effective radiation dose (~2 mSv compared to 3-10 mSv) compared to CTPA.2
Echocardiography
In haemodynamically unstable patients who are unsuitable for CTPA, echocardiography is recommended to aid the diagnosis of a suspected PE.2
Echocardiography may show signs of right ventricular dysfunction that is suggestive of PE, including:
- Abnormal right ventricular ejection pattern – 60/60 sign
- McConnel sign – reduced right ventricular free wall contractility compared with the apex
- Right ventricular dilation and hypokinesis
Negative echocardiography does NOT exclude a PE. A 40-50% negative predictive value for PE has been reported.2
Management
General approach
Acutely unwell patients require an ABCDE approach.
If clinical suspicion of PE is low, the pulmonary embolism rule-out criteria should be used.12
If PE is suspected, perform a two-level PE Wells score without using pulmonary embolism rule-out criteria. While NICE (The National Institute for Health and Care Excellence) guidelines advocate using the Wells score to aid diagnosis, ESC (European Society of Cardiology) guidelines advocate the revised Geneva rule.
Pulmonary Embolism Rule-out Criteria (PERC)
The PERC rule should only be used if the clinical suspicion for PE is low (based on medical history, examination, and investigations) to avoid the overuse of diagnostic testing for PE.12
The PERC rule states that if ALL the following criteria are ABSENT, it reduces the probability of PE to < 2%.12 In such cases, an alternative diagnosis should be considered.
- Age ≥ 50 y/o
- HR ≥ 100 bpm
- SpO2 < 95% on room air
- Unilateral leg swelling
- Haemoptysis
- Recent surgery/trauma ≤4 weeks ago requiring general anaesthesia
- Previous DVT / PE
- Hormone use (oral contraceptives/hormone replacement/estrogenic hormones)
Two-level PE Wells score-guided investigations
Table 1. The Two-level Wells score for PE.12
| Clinical feature | Points |
| Clinical signs and symptoms of DVT (minimum of leg swelling and pain with palpation of the deep veins) | 3 |
| An alternative diagnosis is less likely than PE | 3 |
| Heart rate more than 100 beats per minute | 1.5 |
| Immobilisation for more than 3 days or surgery in the previous 4 weeks | 1.5 |
| Previous DVT/PE | 1.5 |
| Haemoptysis | 1 |
| Malignancy (on treatment, treated in the last 6 months, or palliative) | 1 |
| Clinical probability | |
| PE likely | More than 4 points |
| PE unlikely | 4 points or less |
PE is likely (Wells >4)
Offer immediate computed tomography pulmonary angiogram (CTPA).
If CTPA cannot be done immediately, offer interim therapeutic anticoagulation until a scan is performed.
If CTPA is positive (PE identified):
- Start or continue therapeutic anticoagulation treatment
- If anticoagulation treatment is contraindicated, consider mechanical intervention
If CTPA is negative (PE not identified):
- Consider a proximal leg vein ultrasound scan if DVT is suspected
- If DVT is not suspected → PE is not likely, stop any interim therapeutic anticoagulation but NOT prophylactic anticoagulation for primary prevention and long-term anticoagulation for secondary prevention, consider alternative diagnosis
If PE is likely, why not D-dimer?
Remember, the D-dimer test has low specificity for VTE, making it unsuitable for use in cases where a PE is likely because a positive result does not confirm the diagnosis of PE due to the potential for false positives in other conditions.
PE is unlikely (Wells ≤4)
Offer a D-dimer test with results available within 4 hours.
If the D-dimer test result cannot be obtained within 4 hours, start interim therapeutic anticoagulation.
If the D-dimer test is positive:
- Offer immediate CTPA and give interim therapeutic anticoagulation if there is a delay.
If the D-dimer test is negative:
- PE is not likely
- Stop any interim therapeutic anticoagulation but NOT prophylactic anticoagulation and long-term anticoagulation
- Consider an alternative diagnosis
If PE is unlikely, why use D-dimer?
Remember, the D-dimer test has high sensitivity for VTE, so if the D-dimer is negative PE can be excluded without the patient being exposed to radiation from a CTPA.
Management of confirmed PE
PE with haemodynamic instability
Offer continuous UFH (unfractionated heparin) infusion, and consider thrombolytic therapy:2,12
- IV tissue plasminogen activator (tPA) (e.g. alteplase) is often used
- Catheter-directed thrombolysis
- Open pulmonary embolectomy
Risk assessment (outpatient vs inpatient)
NICE suggest that outpatient treatment for low-risk PE can be considered, using a validated risk stratification tool to determine suitability:12,14
- NICE guidelines did not include a specific tool or scoring system.
- The British Thoracic Society (BTS) guidelines support the use of the Pulmonary Embolism Severity Index (PESI) score
A key requirement for outpatient treatment is being haemodynamically stable.
Patients with intermediate-risk or high-risk PE should be considered for in-patient treatment.
Anticoagulation
In haemodynamically stable PE, management involves anticoagulation therapy:2,12
- First line: apixaban or rivaroxaban, both direct oral anticoagulants (DOACs)
- Second line: low molecular weight heparin (LMWH) followed by dabigatran/edoxaban OR LMWH followed by warfarin
The choice of anticoagulants varies in specific patient populations:2,12,14
- Pregnant: LMWH only (DOACs and warfarin contraindicated)
- Active cancer: DOAC preferred over LMWH
- Severe renal impairment (eGFR < 15 mL/min/1.73m2): UFH or dose-adjusted LMWH
- Antiphospholipid syndrome (triple positive): initial LMWH and warfarin followed by warfarin monotherapy
- All forms of anticoagulation contraindicated: inferior vena cava (IVC) filter
NICE recommends that all patients should have anticoagulation for at least three months. Further anticoagulation is based on the patient’s risk of VTE recurrence and risk of bleeding (ORBIT score is recommended to help assess the risk of bleed).12
As a rule of thumb:12
- If VTE was provoked → 3 months of anticoagulation
- If VTE was unprovoked → 6 months of anticoagulation
- If there is active cancer → 3-6 months of anticoagulation
Provoked vs unprovoked PE
Provoked PE is associated with a recent (within three months) and transient major risk factor (there is no clear-cut guidance, but NICE CKS give examples of significant immobility, surgery, trauma, pregnancy or puerperium, or use of hormonal therapy).12
Unprovoked PE occurs in the absence of a recent (within three months) major clinical risk factor in a person who is not using hormonal therapy.12
It is worth noting that the ESC guidelines no longer support using the terms ‘provoked’ and ‘unprovoked’. They state that they are potentially misleading and unhelpful when making a decision about the duration of anticoagulation.2
Complications
Short-term complications of PE include:
- Sudden cardiac arrest or death can result from ventricular collapse due to massive embolism and occlusion of the pulmonary vasculature
- Pulmonary infarction due to obstruction of arterial blood supply, more common when distal arteries are occluded
- Right ventricular infarction, often secondary to pulmonary hypertension and haemodynamic overload
- Atelectasis
- Exudative pleural effusion
Long-term complications of PE include:3,4
- Increased risk of recurrence, history of previous VTE is a significant risk factor for future VTEs
- Chronic thromboembolic pulmonary hypertension (CTEPH) occurs where fibrotic tissue replaces residual emboli causing chronic obstruction of the pulmonary vasculature, and hence pulmonary hypertension
CTEPH should be excluded in patients with persistent dyspnoea and poor physical performance three to six months after a PE.3,4
Key points
- Pulmonary embolism (PE) results from a dislodged thrombus occluding the pulmonary vasculature. It is potentially life-threatening if not appropriately identified and treated.
- PE shares the same risk factors as DVT, and they can be classified according to Virchow’s triad (venous stasis, hypercoagulable state, endothelial injury).
- Clinical features of PE are highly non-specific; an acute onset makes it more likely, and dyspnoea and tachypnoea are considered the most common presenting features.
- When a PE is suspected, the two-level PE Wells score guides the diagnostic and management approach.
- CT Pulmonary Angiogram (CTPA) is considered the gold standard diagnostic tool.
- The mainstay management of PE involves anticoagulation therapy, usually direct oral anticoagulants (DOACs) for at least 3 months.
- Massive PE or PE with haemodynamic instability carries a high mortality and should be treated aggressively with thrombolytic agents.
Reviewers
Dr Joelle Azzopardi
Respiratory consultant
Dr Adriano Buontempo
Lecturer in Clinical Communication Skills
Editor
Dr Chris Jefferies
References
- Kumar V, Abbas AK, Aster JC, Perkins JA. Chapter 4 Haemodynamic Disorders, Thromboembolism, and Shock. Robbins basic pathology. 10th ed. Philadelphia, PA: Elsevier; 2018.
- Konstantinides SV, Meyer G, Becattini C, Bueno H, Geersing G-J, Harjola V-P, et al. 2019 ESC guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). European Heart Journal. 2019 Aug 31;41(4):543–603.
- BMJ Best Practice. Pulmonary embolism. Reviewed 2023 Oct; updated 2022 Aug. Available from: [LINK]
- NICE Clinical Knowledge Summaries. Pulmonary embolism. Revised 2023 Sep. Available from: [LINK]
- Turetz M, Sideris A, Friedman O, Triphathi N, Horowitz J. Epidemiology, Pathophysiology, and natural history of pulmonary embolism. Seminars in Interventional Radiology. 2018 Jun 4;35(02):92–8.
- Sadeghi A, Brevetti GR, Kim S, Burack JH, Genovese MH, Distant DA, Kodavatiganti R, Lowery RC. Acute massive pulmonary embolism: role of the cardiac surgeon. Tex Heart Inst J. 2005;32(3):430-3.
- Jaff MR, McMurtry MS, Archer SL, Cushman M, Goldenberg N, Goldhaber SZ; et al. (2011). “Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011 Apr 16;123(16):1788-1830.
- RCEM Learning. Pulmonary Embolism. Published in 2023 May 18. Available from: [LINK]
- Walls R, Hockberger R, Gausche-Hill M, Erickson TB, Wilcox SR. Chapter 74 Pulmonary Embolism and Deep Vein Thrombosis. Rosen’s Emergency Medicine: Concepts and Clinical Practice 10th Elsevier Health Sciences; 2022
- Jabaz DF. Pulmonary infarction: Radiology reference article. Radiopaedia.org; 2022. Available from: [LINK]
- Burns E, Buttner R, Buttner EB and R. ECG changes in pulmonary embolism. Published in 2021 Nov 30. Available from: [LINK]
- National Institute for Health and Care Excellence. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing (NICE guideline [NG158]). NICE 2020. Updated 2023 Aug. Available from: [LINK]
- Linkins L‐A., Takach Lapner S. Review of d‐dimer testing: Good, bad, and ugly. International Journal of Laboratory Hematology. 2017 Apr 26;39(S1):98–103.
- Howard LSGE, Barden S, Condliffe R, et al. British Thoracic Society Guideline for the initial outpatient management of pulmonary embolism (PE)Thorax 2018 Jun 13;73:ii1-ii29.
Image references
- Figure 1. Bakerstmd.Saddle thromboembolism. License: [CC BY-SA]
- Figure 2. James Heilman, MD. SaddlePE. License: [CC BY-SA]